Nanodiamonds for Improving Lithium Battery Performance
Nano-diamonds are diamonds that are present in the form of nanoparticles exhibiting that their size is so small that they cannot be seen with the naked eye and rather a microscope is used to see them.
A specialized process with a lot of critical steps is conducted to carry out this formation successfully. Nano-diamonds have a lot of specialized properties that can define their characteristics fully and exhibit these properties in full form through a wide range of applications that these nano-diamonds are capable of exhibiting.Their specialized applications are well-founded in lithium batteries as the range is wide and the area of flourishment just increases for them. Additionally, Nanografi's nano-diamond products and solutions offers a value proposition of enhanced performance and longevity, promising to revolutionize energy storage solutions.
Introduction
This article will provide a thorough examination of nano-diamonds, their applications, and technological advancements in lithium batteries. When they were first found, the general public did not pay them much attention. But as time went on and technology advanced, these tiny diamonds proved to be valuable for a wide range of uses and advantages in the post-revolutionary world. As we go more into its characteristics, we will discover that these seemingly simple little particles are very complex and contain some components that are singular from their point of view. To analyze these constituents and then merge them into an entirely new set of particles has been a lengthy and arduous journey. Today, lithium batteries are a vital component of modern technology.
History of Nanodiamonds
The first nanoscale diamond particles were created by blasting in the USSR in the 1960s, but the rest of the world was mostly unaware of them until the late 1980s. Following that, a string of significant discoveries made starting in the late 1990s stimulated interest in tiny particles, now known as nanodiamonds.
1) Single-digit nanodiamond colloidal solutions with a diameter of 4-5 nm are now commercially available for the first time.
2) Fluorescent nanodiamonds are now being used by scientists in place of semiconductor quantum dots for biological imaging since they are non-toxic.
3) Third, diamond-based nanoscale magnetic sensors have been created.
4) Fourth, a variety of wet gas chemistry techniques can be employed to characterize the characteristics of nanodiamonds thanks to their chemical reactivity.
The development of new environmentally friendly refining techniques has made it possible to make high-purity nanodiamond powders in large quantities at low cost and with controlled surface chemistry.
6) Finally, nanodiamonds are being researched for use in biomedical imaging, drug delivery, and other medical procedures since they are less harmful than other carbon nanoparticles.
Model Building of Nanodiamonds
The presence and variety of surface functional groups, sp2 carbon, and particle shape must be taken into account while creating models of nanodiamonds. Nanodiamond particles that have been thoroughly cleansed have almost flawless crystal structures with hardly any non-diamond carbon. According to observations made using transmission electron microscopy (18,19,46), the nanodiamond particles either have an amorphous carbon core with dangling bonds terminated with functional groups or a diamond core made of sp3 carbon that is partially covered by a graphite shell.
Nitrogen impurities (up to 2-3 wt%) that can form complexes in the core of the nanodiamond particles, the presence of twins, and grain boundaries are additional characteristics of nanodiamonds that are not taken into account by this model. microcrystals. The latter may be to blame for the X-ray diffraction peaks that were previously attributed to the sp3-disordered carbons being broader.
Mechanism of Nanodiamond Formation
A mechanism for how nanodiamonds develop during explosion was put forth by Danilenko. Although the Juguet point's pressure and temperature are too low to create bulk liquid carbon, they are high enough to create liquid carbon at the nanoscale. The nanodiamond stability regime shifts slightly to higher pressures for nano carbons, while the liquid carbon regime shifts to lower temperatures. As a result, it is presumable that liquid carbon condenses and crystallizes to form homogeneous nanodiamonds in a volume of supersaturated carbon vapor.
Other explosives-based processes, such as creating nanodiamonds from graphite using shock waves, result in nanodiamonds with crystallite sizes larger than 10 nm and non-combustible impurities (metals and oxides, 1–8 wt%). Probably require cleaning. The used igniters (often lead, silver, or copper azides) contain metal impurities.
Vibrational Spectroscopy of Nanodiamond
Understanding the phase composition and surface terminations of nanodiamonds is made possible through Raman and Fourier transform infrared (FTIR) spectroscopy. In addition to detecting functional groups and molecules adsorbed on surfaces, FTIR may also spot changes in the surface chemistry of functionalized nanodiamonds 5,8,14,79. In nanodiamonds, nitrogen detects appear as two broad bands overlapping with the peaks of surface functional groups in the range 1,100–2,500 cm-1.

Figure 1. Structure of nanodiamond.
Properties of Nanodiamonds
The majority of the superior qualities of bulk diamonds are carried over by nanodiamonds, which provide them at the nanoscale. Diamond is a superior material in many ways. Excellent hardness and modulus, biocompatibility, optical qualities, resistivity against thermal and electrical activities, stability during chemical reactions, and the resistance that may be encountered by nano-diamonds which might cause environmental changes and alter a few of these characteristics.
Fluorescence
The beneficial fluorescence features of nanodiamonds are provided by the presence of nitrogen-vacancy (NV) centers, which are nitrogen atoms close to vacancies.
By blasting nanodiamonds with energetic particles (in the form of electrons, protons, and helium ions), vacuum annealing takes place at 600–800 °C can produce NV centers. Diamond develops vacancies as a result of irradiation, and during the process of annealing, these vacancies are entitled to move and are then caught by nitrogen atoms which are usually present in the diamond itself.
Two Types of NV Centers
Neutral (NV0) and negatively charged (NV-) NV centers are generated, and their emission spectra are dissimilar. Because the NV-center possesses an S = 1 spin ground state that may be spin-polarized by optical pumping and controlled by electron paramagnetic resonance, it is particularly interesting. The spin coherence time is also very long. Nanodiamond NV centers are also being researched for applications in high-resolution magnetic sensing, fluorescence resonance energy transfer, and biomedical imaging.
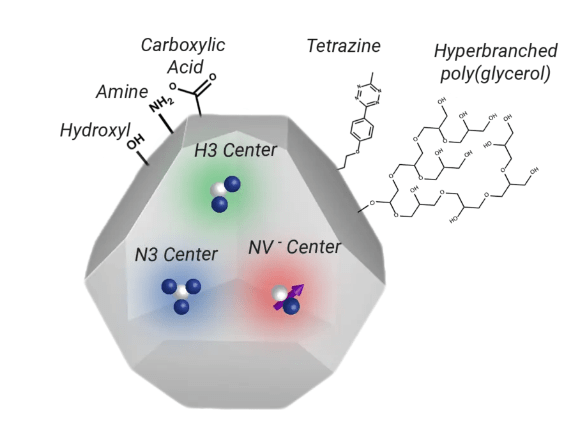
Figure 2. Functionalized multicolor fluorescent nanodiamond.
Biocompatibility and Fate in the Body of Nanodiamonds
Although glassy carbon and diamond are known to be non-toxic substances, we cannot infer the same for carbon nanoparticles. The toxicity of nanodiamonds is a legitimate concern since different manufacturers employ various purifying techniques, and there are several choices for surface modification. Studies have been done both in vitro and in vivo to look at a variety of traits, including cell survival, gene program activities, and in vivo mechanistic and physiological behavior.
Nanodiamonds in Trachea
The presence of nanodiamonds in the alveolar region is certainly decreasing as time is passing by and the macrophages that are present are bound to be loaded with nanodiamonds. This enables their presence visually more active in the bronchia for 28 days after the exposure has occurred. The final report conducted that the nanodiamonds present in the trachea comprise minimal pulmonary toxicity.
The high doses of nanodiamond complexes given intravenously did not affect the serum markers of hepatic and systemic toxicity.
Characterizing the Spectral Features
Nano-diamonds have a lot of characteristics and features after the process of oxidative purification has been conducted which includes the stretching of O-H as it occupies 3200 to 3600 cms and the bending includes 1630 to 1640 cms in which the bands are originated from both the species of nano-species and their surfaces. In this case, C=O can become a part of ketone, aldehyde, a few forms of carboxylic acids, ester, and some other forms of materials that can be a part of nano-diamonds.
Broad Absorption Feature
Additionally, many nanodiamonds have a very broad absorption characteristic with multiple overlapping peaks between 1,000 and 1,500 cm-1, commonly referred to as the "fingerprint zone." O-H bends, C-O-C stretches, epoxy C-O stretches, C-C stretches, amide C-N stretches, and C-N-H bends are a few examples spikes brought on by deficiencies in nitrogen. NO2, SO2OH, and other groups vibrate.
Availability of Nanodiamonds for Research
Today, a dizzying array of nanodiamonds are available for investigation. They were made using detonation techniques, laser ablation, high-energy ball milling of HPHT diamond crystallites, plasma-enhanced chemical vapor deposition (CVD), autoclave synthesis from ultracritical fluids, chlorination of carbides, ion bombardment of graphite, electron bombardment of carbon onions, ultrasonic cavitation, and the first three of these techniques are currently used in industry. Astronomical evidence suggests that nanodiamonds are present in the protoplanetary discs of specific types of stars, while the origin of these cosmic sources is still being researched. For research and industrial usage, we now need to be able to mass-create nanodiamonds on Earth. Here, we cover the production, characteristics, and uses of nanodiamonds, which range in size from 2 to 10 nm.
Graphite: The Stable Form of Carbon
At room temperature and pressure, graphite is the most stable form of carbon, although diamond is known to be metastable. Although the energy barrier between the two phases is rather large (about 0.4 eV per atom), there is still a need for high temperatures, high pressure, and/or catalysts because of the energy difference between the two phases. However, the carbon phase diagram should also take cluster size into account as a third parameter since, at the nanoscale, the Gibbs free energy is dependent on the surface energy, which affects the phase diagram (besides pressure and temperature).
Stability of Different Carbon Phases
Numerous theoretical and computational studies have examined the stability of various nanoscale carbon phases. Tetrahedral hydrocarbons are more stable than polyaromatics with diameters less than 3-6 nm, according to atomistic models. Later research 41 revealed that shape has a significant impact on surface reconstruction and sp2 carbon production, contributing to the stability of nanodiamonds.
The surfaces of the octahedrons, cubo-octahedrons and globular clusters display transitions from sp3 to sp2 carbons, whereas the naked (unfunctionalized) surfaces of the cubic crystals display a structure resembling that of a bulk diamond.
Preferential Exfoliation
Starting with sub-nanometer clusters, preferential exfoliation of the surface encourages cluster transitions to endofullerenes for small clusters (tens of atoms) and an onion-like shell with a diamond core ("bucky diamond") for bigger clusters. cluster (hundreds of atoms) (hundreds of atoms) 41; Large, irregular clusters (1-3.3 nm), with thin (monolayer or bilayer) graphite shells, are expected to remain stable within the diamond structure and have less than 76% surface graphitization.
Cluster Surface
It is recommended that functional group terminations or the conversion to sp2 carbons stabilize the surface of the sp3 cluster. As a result, surface termination as well as particle size, shape, and stability of carbon nanoparticles. Only the function of hydrogen termination has been thoroughly investigated thus far. Oils include stable compounds called diamondoids, which are hydrogen-terminated nanodiamonds with diameters less than 1.5 nm33. When the diamond size reaches 3 nm or more, first-principles simulations43 demonstrate that bucky diamonds have an energy advantage over hydrogenated nanodiamonds.
Results
According to preliminary findings from modeling nanodiamonds with functional groups that comprise oxygen (group 44) and nitrogen (group 45), different groups have distinct nanodiamond facets depending on temperature and particle environment, nanodiamond size, and morphology.
To learn about thermal management potential of graphite sheets
in technology and industry, read our blog.
Current and Future Applications of Nanodiamonds for Improving Lithium Battery Performance
Nanodiamond additions have been utilized in electrolytic and electroless plating for a very long time. It has also been employed more recently in applications like mass spectrometry, 105 chemical vapor deposition of diamond films, 106 chromatography, 52,107 proteomics, and magnetic resonance imaging. It is demonstrated that carbon "onions" are made via the process of graphitization of nanodiamonds which tend to be used in energy storage, composites, and catalysis reactions (nanodiamonds also tend to exhibit catalytic activity). Electrochemical double-layer capacitors, batteries, and electrical analysis are all possible uses for boron-doped nanodiamonds.
Undoped Insulating Nanodiamonds
Additionally, in electrochemical environments, undoped insulating nanodiamonds display redox activity. Applications in five tribology and lubrication domains are the main topics of this section. Drug delivery, tissue scaffolds, protein mimics, nanocomposites, and surgical implants are a few examples. Due to nanodiamonds' ability to self-assemble and their ability to bind a range of tiny molecules, proteins, antibodies, medicines, and nucleic acids to their surface, the final three of these applications are made conceivable.
To learn more about the applications of silicon anode materials for lithium batteries,
you can read our blog post here.
Nanodiamonds as Electrolyte Additives
Batteries using lithium ions (LIB). Li+/Li has a redox potential of -3.04 V (versus. high working voltage is provided with a full cell. When lithium metal anodes and high-capacity cathode materials are coupled to create complete cells, these characteristics offer great energy density. Rechargeable lithium-metal batteries (LMBs), including lithium-sulfur (Li-S) and lithium-oxygen (Li-O2) batteries, are therefore viewed as attractive contenders for high energy density storage.
Dendrites of lithium, However, LMB can result in harmful Li dendrites, which restricts its usefulness and can cause cell shorting, thermal runaway, and failures that could cause an explosion or fire. Li dendrites expand the surface area where Li metal and organic electrolytes come in contact and encourage side reactions. Lower coulombic efficiency results from the reaction products' electronic separation of the Li metal from the conducting matrix.
Suppression of Lithium Dendrite Growth
Although there has been a lot of research on how to stop lithium dendrite growth, there hasn't been as much done to understand how lithium metal grows and forms nuclei. Cui and his coworkers recently investigated Li's potential for nucleation on various current collectors. Their findings showed substrate-dependent nucleation behavior due to the selective deposition of Li metal on chosen substrates that they accomplished.
Nanodiamond electrolytes have stronger polarization, a shorter LMB lifetime, and higher coulombic efficiency.
Low-Cost Nanodiamonds
The nanodiamond particles employed in this study were made using a low-cost, commercial detonation technique, then modified by being covalently attached to ODA37,38 and carboxylated. They have high crystallinity and 5-nm-sized crystals. The interplanar crystallographic spacing, which corresponds to diamond (111) planes (0.206 nm, PDF#65-0537), is measured to be 0.21 nm in lattice-edge transmission electron microscopy (TEM) images. It was created as an EC/DEC electrolyte with modified nanodiamond particles scattered throughout and a saturation concentration of 0.82 mg mL-1. The solution changed color from the colorless, clear EC/DEC electrolyte to a pale yellow tint with the addition of nanodiamond particles. In comparison to pure commercial nanodiamonds, the size of this cluster was substantially smaller39. The size and color distribution of the nanodiamond clusters in the solution in the electrolyte.
Nanodiamond Surface
Among these materials, nanodiamond has the lowest energy barrier for Li diffusion. This shows that the Li ions tend to adsorb on the nanodiamond surface at the cathode (Cu foil)-electrolyte interface, decrease the aggregation, and easily disperse and distribute uniformly, producing a morphology devoid of dendrites.
The Behavior of Electrochemical Cycling
Testing symmetric Li has proven the Li electrode's long-term electrochemical cycling stability. Check out the Li cell. The symmetric Li|Li electrodes demonstrated stable cycling in the nanodiamond electrolyte for 200 hours and 150 hours, respectively, and displayed polarisation (100 mV at 1 mA cm to 120 mV at 2 mA cm-2).
Concentration of Nanodiamonds
The cells exhibited good stability even after voltage application when the nanodiamond concentration was reduced from 0.82 to 0.41 mg mL-1.
The electrolyte with 0.41 mg mL1 nanodiamonds performed significantly better than the electrolyte without them. The cycling stability of the 0.82 mg mL1 electrolyte was comparable, although the voltage polarisation increased more slowly over longer cycles. The importance of nanodiamonds in stabilizing Li metal to obtain stable long-term cycling performance is amply demonstrated by these data.
Modification of Nanodiamond
1.5 g of nanodiamonds were modified from nanodiamond particles and cleaned by air oxidation at 425 °C before being boiled for 24 hours in a solution of HCl, HNO3, and distilled water to eliminate metallic impurities. A known catalyst for this reaction, anhydrous N, N-dimethylformamide, was added to 1.5 g of the resultant material and refluxed for 24 hours at 70 °C with 50 mL of SOCl2 and 1 mL of SOCl2. After distillation to remove the supernatant, the resulting solid was three times washed with anhydrous tetrahydrofuran before being vacuum-dried at room temperature. Then, 5 g of ODA was mixed with the chlorinated nanodiamond powder for 96 hours at 90 to 100 °C in a covered flask.
The dispersion of nanodiamonds in organic solvents is easy. The creation of an electrolyte incorporating nanodiamonds involved dispersing 50 mg of the resulting ODA-functionalized nanodiamond particles in 10 mL of an Ar1-filled glove box.
Conclusion
In light of the above-stated facts, nanodiamonds are elements that have a great tendency to merge themselves into multiple particles and give new forms to themselves which can benefit us in many ways, from being used in batteries to fluorescent bulbs it can have applications that are to be of immense importance to the upcoming era of technology. Hence, it can be stated very safely that nanodiamonds are to exhibit a great role in modernizing our day-to-day life.
To discover the latest news from nanotechnology, you can visit Blografi.
References
4 Great Uses of Carbon Quantum Dots - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/4-great-uses-of-carbon-quantum-dots/
Advantages of Silicon Anode Materials for Lithium-Ion Batteries - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/advantages-of-silicon-anode-materials-for-lithiumion-batteries-/
Chemical Vapor Deposition CVD Graphene - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/chemical-vapor-deposition-cvd-graphene/
Cheng, X. B., Zhao, M. Q., Chen, C., Pentecost, A., Maleski, K., Mathis, T., Zhang, X. Q., Zhang, Q., Jiang, J., & Gogotsi, Y. (2017). Nanodiamonds suppress the growth of lithium dendrites. Nature Communications 2017 8:1, 8(1), 1–9. https://doi.org/10.1038/s41467-017-00519-2
Deng, D. (2015). Li-ion batteries: Basics, progress, and challenges. Energy Science and Engineering, 3(5), 385–418. https://doi.org/10.1002/ESE3.95
Enhancing Thermal Energy Storage with Nanoparticles - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/enhancing-thermal-energy-storage-with-nanoparticles/
Functionalized Multicolor Fluorescent Nanodiamond – Adámas Nanotechnologies. (n.d.). Retrieved April 29, 2024, from https://www.adamasnano.com/functionalized-multicolor-fluorescent-nanodiamond/
Graphite Nanoparticles and Nanopowder - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/graphite-nanoparticles-and-nanopowder/
Graphite Sheet as Brilliant Thermal Interface Material - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/graphite-sheet-as-brilliant-thermal-interface-material/
Kausar, A., Ashraf, R., & Siddiq, M. (2014). Polymer/Nanodiamond Composites in Li-Ion Batteries: A Review. Polymer-Plastics Technology and Engineering, 53(6), 550–563. https://doi.org/10.1080/03602559.2013.854386
Lithium Nickel Cobalt Aluminum Oxide (NCA) in Lithium-Ion Battery Applications - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/lithium-nickel-cobalt-aluminum-oxide-nca-in-lithiumion-battery-applications/
Lithium-Ion Batteries: How They Work, Where They Are Used, Advantages & Disadvantages - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/lithiumion-batteries-how-they-work-where-they-are-used-advantages-disadvantages/
Mochalin, V. N., Shenderova, O., Ho, D., & Gogotsi, Y. (2012). The properties and applications of nanodiamonds. Nature Nanotechnology, 7(1), 11–23. https://doi.org/10.1038/NNANO.2011.209
Mochalin, V., Shenderova, O., Ho, D., & Gogotsi, Y. (2020). The Properties and Applications of Nanodiamonds. Nano-Enabled Medical Applications, 313–350. https://doi.org/10.1201/9780429399039-11
Nanomedicine and Nanodrugs - Nanografi Blog - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/nanomedicine-and-nanodrugs/
Nanoparticles Dispersions - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/nanoparticles-dispersions/
Nitrogen-Containing Graphene Ribbons - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/nitrogencontaining-graphene-ribbons/
Strategies for Improving Rechargeable Lithium-ion Batteries - Nanografi Nano Technology. (n.d.). Retrieved April 29, 2024, from https://nanografi.com/blog/strategies-for-improving-rechargeable-lithiumion-batteries/
Thekkedath, A., Sridharan, K., Thekkedath, A., & Sridharan, K. (2022). Nanodiamonds and Its Applications. Applications and Use of Diamond. https://doi.org/10.5772/INTECHOPEN.108326
Recent Posts
-
Advanced Materials for Unmanned Aerial Vehicle (UAV) Protection Against Laser
Consider a UAV on a critical mission, rendered inoperative by a sudden laser attack. With the increa …26th Jul 2024 -
Simulation and Modeling of Material Properties
Our world is composed of a dazzling array of materials, each with its own unique properties that dic …19th Jul 2024 -
Advanced Coatings for Superior Corrosion and Wear Resistance
Corrosion and wear pose significant challenges across various industries, leading to substantial eco …12th Jul 2024







