Titanium Dioxide Sputtering Targets
Titanium dioxide is the naturally occurring oxide of titanium, chemical formula TiO2. When used as a pigment, it is called titanium white. Generally, it is sourced from ilmenite, rutile and anatase. Titanium dioxide has a wide range of applications, including paint, sunscreen and food coloring.
Sputtering is initiated due to the bombardment of energetic particles at the target. These energetic particles are generally ions. Two approaches can be followed to produce ions and sputter the target materials. The first is quite straightforward by using an ion source which is aimed toward the target. Collecting the sputtered particles on a substrate enables the deposition of a thin film. The sputtering process with the assistant of magnetron cathodes is called magnetron sputtering which is widely used in both scientific and industrial fields. A magnetron uses a magnetic field to confine electrons close to the cathode, making it easier to sustain an electrical discharge at low pressure. There are various geometries but all magnets are arranged in such a way that the magnetic field lines are parallel to the cathode surface and perpendicular to the electric field lines. This causes the secondary electrons to drift in a closed path in the – E ×B direction, also known as the Hall Effect, trapping the electrons near the cathode surface. This arrangement results in enhanced ion bombardment and sputtering rates for both DC and RF discharges. Magnetron sputtering is particularly useful when high deposition rates and low substrate temperatures are required. In RF magnetron sputtering, cathode and anode are changeable and for very short cycles, target functions as anode which cause the removal of insulating layer. By doing so, the process can continue.
Transition metal oxides are often nonstoichiometric, at near-atmospheric oxygen pressure the oxygen vacancies is the predominant defect in titanium oxide. The oxygen deficiency introduces an excess of electrons in the material resulting in an increase of the electrical conductivity. The oxygen vacancies act as electron donors, thus titanium dioxide is an n-type semiconductor, in contrast with p-type semiconductors which contain electron acceptors and where the charge carriers are holes rather than electrons.
Nano-crystalline titanium dioxide is one of the most investigated oxide materials due to its high refractive index, high dielectric constant, and optical transmittance in the visible and near-IR region. Titanium dioxide has been attracted a lot of interest for a wide range of applications, such as dye-sensitized solar cells, photo catalysts, optical coatings and capacitors for large-scale integrated (LSI) devices. Thin films and nanostructures based on titanium dioxide are extensively studied for various applications because of their multifunctional nature. Among MOS (metal oxide semiconductors), the stability of titanium dioxide in harsh environment makes it a good candidate for gas sensing applications.
- Product
- Qty in Cart
- Quantity
- Price
- Subtotal
-
Titanium Dioxide (TiO2) Sputtering Targets, indium, Purity: 99.99%, Size: 4'', Thickness: 0.125'', Beige to White
€2,002.001 piece/2002 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets, indium Purity: 99.99%, Size: 4'', Thickness: 0.125'', Beige to White Sputtering is a proven...NG0ST01TI80€2,002.00 -

Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.99%, Size: 3'', Thickness: 0.125'', Beige to White
€1,430.001 piece/1430 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.99%, Size: 3'', Thickness: 0.125'', Beige to White Sputtering is a proven technology capable of depositing thin...NG0ST01TI79€1,430.00 -
Titanium Dioxide (TiO2) Sputtering Targets, indium, Purity: 99.99%, Size: 2'', Thickness: 0.125'', Beige to White
€980.001 piece/980 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets, indium Purity: 99.99%, Size: 2'', Thickness: 0.125'', Beige to White Sputtering is a proven technology capable of...NG0ST01TI78€980.00 -
Titanium Dioxide (TiO2) Sputtering Targets, indium, Purity: 99.99%, Size: 1'', Thickness: 0.125'', Beige to White
€823.001 piece/823 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets, indium Purity: 99.99%, Size: 1'', Thickness: 0.125'', Beige to White Sputtering is a proven technology capable of...NG0ST01TI77€823.00 -
Titanium Dioxide (TiO2) Sputtering Targets, indium, Purity: 99.95%, Size: 4'', Thickness: 0.125'' , Grey to Black
€909.001 piece/909 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets, indium Purity: 99.95%, Size: 4'', Thickness: 0.125'' , Grey to Black Sputtering is a proven technology...NG0ST01TI76€909.00 -
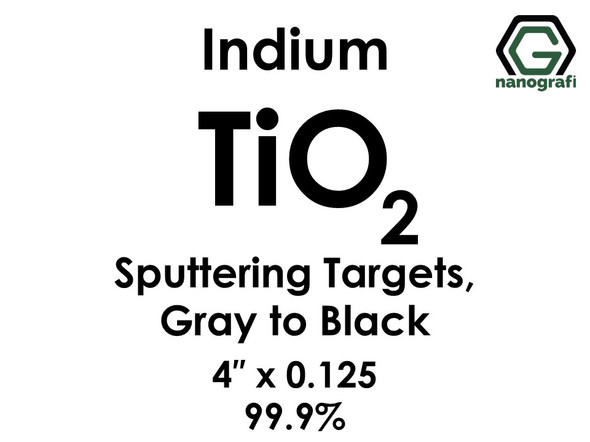
Titanium Dioxide (TiO2) Sputtering Targets, indium, Purity: 99.9%, Size: 4'', Thickness: 0.125'', Grey to Black
€986.001 piece/986 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets, indium Purity: 99.9%, Size: 4'', Thickness: 0.125'', Grey to Black Sputtering is a proven technology capable of depositing...NG0ST01TI75€986.00 -
Titanium Dioxide (TiO2) Sputtering Targets, indium, Purity: 99.9%, Size: 3'', Thickness: 0.125'', Grey to Black
€928.001 piece/928 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets, indium Purity: 99.9%, Size: 3'', Thickness: 0.125'', Grey to Black Sputtering is a proven technology capable of...NG0ST01TI74€928.00 -
Titanium Dioxide (TiO2) Sputtering Targets, elastomer, Purity: 99.9%, Size: 2'', Thickness: 0.125'', Grey to Black
€594.001 piece/594 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets, elastomer Purity: 99.9%, Size: 2'', Thickness: 0.125'', Grey to Black Sputtering is a proven technology capable of depositing...NG0ST01TI73€594.00 -
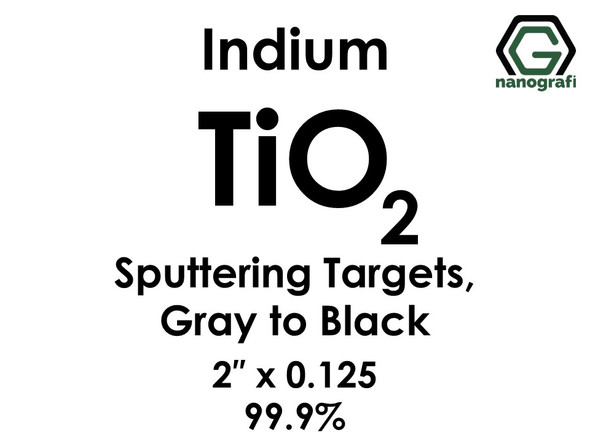
Titanium Dioxide (TiO2) Sputtering Targets, indium, Purity: 99.9%, Size: 2'', Thickness: 0.125'', Grey to Black
€624.001 piece/624 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets, indium Purity: 99.9%, Size: 2'', Thickness: 0.125'', Grey to Black Sputtering is a proven technology capable of...NG0ST01TI72€624.00 -
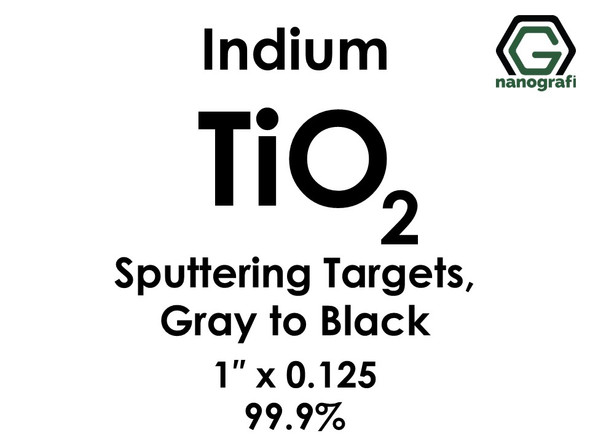
Titanium Dioxide (TiO2) Sputtering Targets, indium, Purity: 99.9%, Size: 1'', Thickness: 0.125'', Grey to Black
€506.001 piece/506 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets, indium Purity: 99.9%, Size: 1'', Thickness: 0.125'', Grey to Black Sputtering is a proven technology capable of depositing...NG0ST01TI71€506.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.99%, Size: 8'', Thickness: 0.250'', Beige to White
€1,608.001 piece/1608 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.99%, Size: 8'', Thickness: 0.250'', Beige to White Sputtering is a proven technology capable of depositing...NG0ST01TI70€1,608.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.99%, Size: 6'', Thickness: 0.250'', Beige to White
€1,620.001 piece/1620 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.99%, Size: 6'', Thickness: 0.250'', Beige to White Sputtering is a proven technology capable of...NG0ST01TI69€1,620.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.9%, Size: 6'', Thickness: 0.250'', Grey to Black
€650.001 piece/650 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.9%, Size: 6'', Thickness: 0.250'', Grey to Black Sputtering is a proven technology capable of depositing thin...NG0ST01TI68€650.00 -
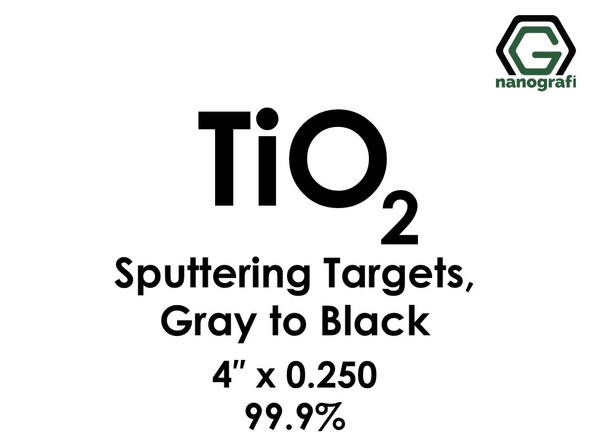
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.9%, Size: 4'', Thickness: 0.250'', Grey to Black
€542.001 piece/542 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.9%, Size: 4'', Thickness: 0.250'', Grey to Black Sputtering is a proven technology capable of depositing...NG0ST01TI66€542.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.99%, Size: 4'', Thickness: 0.250'', Beige to White
€1,751.001 piece/1751 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.99%, Size: 4'', Thickness: 0.250'', Beige to White Sputtering is a proven technology capable of depositing...NG0ST01TI67€1,751.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.99%, Size: 4'', Thickness: 0.125'', Beige to White
€1,544.001 piece/1544 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.99%, Size: 4'', Thickness: 0.125'', Beige to White Sputtering is a proven technology capable of...NG0ST01TI65€1,544.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.9%, Size: 4'', Thickness: 0.125'', Grey to Black
€526.001 piece/526 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.9%, Size: 4'', Thickness: 0.125'', Grey to Black Sputtering is a proven technology capable of depositing thin...NG0ST01TI64€526.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.99%, Size: 3'', Thickness: 0.250'', Beige to White
€1,176.001 piece/1176 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.99%, Size: 3'', Thickness: 0.250'', Beige to White Sputtering is a proven technology capable of...NG0ST01TI63€1,176.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.9%, Size: 3'', Thickness: 0.250'', Grey to Black
€506.001 piece/506 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.9%, Size: 3'', Thickness: 0.250'', Grey to Black Sputtering is a proven technology capable of...NG0ST01TI62€506.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.9%, Size: 3'', Thickness: 0.125'', Beige to White
€980.001 piece/980 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.9%, Size: 3'', Thickness: 0.125'', Beige to White Sputtering is a proven technology capable of depositing thin...NG0ST01TI61€980.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.9%, Size: 3'', Thickness: 0.125'', Grey to Black
€476.001 piece/476 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.9%, Size: 3'', Thickness: 0.125'', Grey to Black Sputtering is a proven technology capable of depositing thin...NG0ST01TI60€476.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.99%, Size: 2'', Thickness: 0.250'', Beige to White
€848.001 piece/707 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.99%, Size: 2'', Thickness: 0.250'', Beige to White Sputtering is a proven technology capable of...NG0ST01TI59€848.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.9%, Size: 2'', Thickness: 0.250'', Grey to Black
€392.001 piece/392 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.9%, Size: 2'', Thickness: 0.250'', Grey to Black Sputtering is a proven technology capable of depositing thin...NG0ST01TI58€392.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.99%, Size: 2'', Thickness: 0.125'', Beige to White
€588.001 piece/588 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.99%, Size: 2'', Thickness: 0.125'', Beige to White Sputtering is a proven technology capable of...NG0ST01TI57€588.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.9%, Size: 2'', Thickness: 0.125'', Grey to Black
€349.001 piece/349 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.9%, Size: 2'', Thickness: 0.125'', Grey to Black Sputtering is a proven technology capable of depositing thin...NG0ST01TI56€349.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.99%, Size: 1'', Thickness: 0.250'', Beige to White
€705.001 piece/705 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.99%, Size: 1'', Thickness: 0.250'', Beige to White Sputtering is a proven technology capable of...NG0ST01TI55€705.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.9%, Size: 1'', Thickness: 0.250'', Grey to Black
€282.001 piece/282 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.9%, Size: 1'', Thickness: 0.250'', Grey to Black Sputtering is a proven technology capable of...NG0ST01TI54€282.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.99%, Size: 1'', Thickness: 0.125'', Beige to White
€556.001 piece/556 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.99%, Size: 1'', Thickness: 0.125'', Beige to White Sputtering is a proven technology capable of...NG0ST01TI53€556.00 -
Titanium Dioxide (TiO2) Sputtering Targets, Purity: 99.9%, Size: 1'', Thickness: 0.125'', Grey to Black
€241.001 piece/241 € Please contact us for quotes on larger quantities !!! Titanium Dioxide (TiO2) Sputtering Targets Purity: 99.9%, Size: 1'', Thickness: 0.125'', Grey to Black Sputtering is a proven technology capable of depositing...NG0ST01TI52€241.00