Magnesium Oxide (MgO) Nanoparticles: Synthesis, Properties and Applications
In the realm of nanotechnology, magnesium oxide (MgO) nanoparticles have emerged as a subject of immense interest due to their unique physical and chemical properties. These nanoparticles, with dimensions measured in nanometers, exhibit characteristics that are not found in their bulk counterparts, leading to potential applications in various fields including catalysis, energy, and environmental remediation.
The burgeoning research into MgO nanoparticles is driven by their high thermal stability, excellent dielectric properties, and robustness as an antioxidant, which make them a versatile material for technological advancements. Nanografi's magnesium oxide nanoparticles are engineered to meet the highest standards, providing researchers and technologists with a reliable and versatile nanomaterial that drives innovation and efficiency across multiple applications.
Introduction
Magnesium oxide nanoparticles possess a cubic crystal structure and are known for their high melting point, making them suitable for applications that require thermal resistance. Furthermore, their ability to act as a base, absorbing acids and toxic gases, positions them as a promising material for environmental cleanup and protection. The surface of MgO nanoparticles can be engineered to enhance their reactivity and interaction with other substances, opening up avenues for their use in catalysis and as adsorbents in the removal of pollutants from air and water.
Structure of Magnesium Oxide Nanoparticles
Magnesium oxide (MgO) nanoparticles are ultra-small particles, usually composed of magnesium and oxygen atoms. They are characterised by their size on the nanometre scale (typically between 1 and 100 nanometres). MgO nanoparticles have a cubic crystal structure, with each magnesium ion octahedrally coordinated by six oxygen ions. This structure contributes to the properties of MgO such as high melting point, mechanical strength and chemical resistance.

Figure 1. Crystal structure of Magnesium Oxide nanoparticles.
Synthesis of Magnesium Oxide Nanoparticles
Nanoparticles themselves are typically synthesized through various chemical methods that allow for precise control over their size and shape, factors which directly influence their chemical reactivity and physical properties. Techniques such as sol-gel processes, hydrothermal synthesis, and combustion methods are commonly employed to produce these nanoparticles with high purity and uniformity.
Here, three common techniques used in the synthesis of magnesium oxide nanoparticles are reviewed: sol-gel processes, hydrothermal synthesis and combustion methods.
Sol-Gel Process: This involves using magnesium salts as precursors, which are hydrolyzed and then undergo condensation to form a gel. The gel is dried and calcined at high temperatures to produce fine, crystalline MgO nanoparticles.
Hydrothermal Synthesis: Magnesium salts are dissolved in water under high pressure and temperature in an autoclave. This process leads to the formation of magnesium hydroxide, which then converts into MgO upon cooling or further heating, allowing for control over particle size and crystallinity.
Combustion Synthesis: A mixture of a magnesium precursor and an organic fuel is ignited, leading to a rapid, exothermic reaction that produces MgO directly. This method is fast and suitable for large-scale production but results in more porous particles.
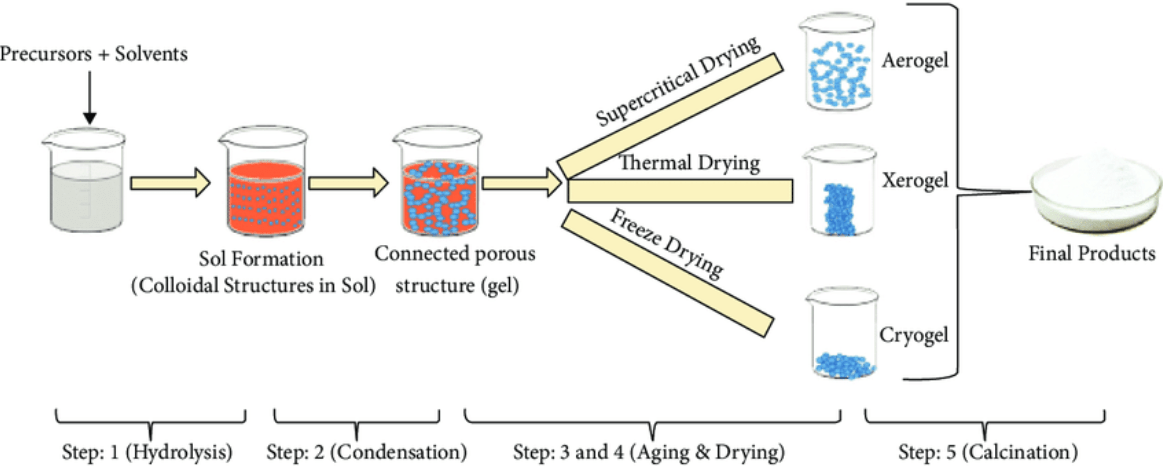
Figure 2. Schematic of different stages of sol-gel process: from precursor to aerogel [1-5].
Properties of Magnesium Oxide Nanoparticles
Magnesium oxide nanoparticles are distinguished by their extremely small size, typically under 100 nanometers. These nanoparticles possess unique properties that differentiate them significantly from their bulk material counterparts.
Enhanced Surface Area and Reactivity
A pivotal property of magnesium oxide nanoparticles is their exceptionally high surface area-to-volume ratio. This structural attribute significantly enhances their reactivity, making them potent catalysts in various chemical reactions. Their ability to accelerate reaction rates efficiently and effectively, without being consumed, is a key advantage in industrial applications.
Did you know that platinum nanoparticles are used in catalyst applications for fuel cells? Learn now.
High Thermal Stability
One of the most important properties of magnesium oxide nanoparticles is their ability to withstand very high temperatures without breaking down or changing in form. This makes them perfect for use in environments that get extremely hot, such as in furnaces used in industrial settings, or as protective coatings that can resist heat.
Excellent Dielectric Properties
Magnesium oxide nanoparticles are also great insulators, meaning they can prevent the flow of electrical current, making them essential in electronic devices. They help to maintain the safety and efficiency of electronic components by ensuring that electricity is directed where it is needed and not where it could cause damage or inefficiency.
Robustness as an Antioxidant
Magnesium oxide nanoparticles are valued for their antioxidant properties, which help combat oxidative stress by neutralizing damaging free radicals. This action extends the lifespan and enhances the performance of various materials, including plastics, by protecting them from environmental degradation.
Additionally, advancements in green synthesis methods for these nanoparticles have not only reduced environmental impact but also increased their biomedical applications. Notably, these eco-friendly nanoparticles show promising anticancer properties, demonstrating their dual utility in both material science and medical research.
Applications of Magnesium Oxide (MgO) Nanoparticles
Magnesium oxide (MgO) nanoparticles are an incredibly versatile nanomaterial with a wide range of applications across various sectors, from environmental management to high-tech industries. These nanoparticles are not only effective in enhancing product performance but also play a crucial role in advancing sustainable and safe technologies.
Here, we examine the various uses of MgO nanoparticles in important industries, revealing their importance and the benefits they bring to each field.
Environmental Applications
Magnesium oxide (MgO) nanoparticles are highly valued in environmental technology, particularly in water treatment processes. Their high adsorption capacity allows them to effectively remove pollutants such as heavy metals and dyes from wastewater. This makes MgO nanoparticles an eco-friendly solution for maintaining clean water supplies and supporting sustainable environmental practices.

Figure 3. Schematic representation of the use of magnesium oxide in water treatment.
Industrial Uses
In the industrial sector, the thermal and chemical stability of magnesium oxide nanoparticles enhances the performance of refractory materials used in high-temperature processes, such as steel and glass production. Additionally, their ability to act as a flame retardant makes them useful in developing fire-resistant materials, contributing significantly to safety in construction and manufacturing.
Healthcare and Medicine
Magnesium oxide (MgO) nanoparticles have promising applications in the medical field due to their antibacterial properties. They are used in coatings for medical devices to prevent infections, and in wound dressings to accelerate healing by reducing bacterial growth. Moreover, their non-toxicity and biocompatibility make them suitable for drug delivery systems, where they can safely transport therapeutic agents to targeted areas within the body.
Electronics and Energy
In electronics, the excellent dielectric properties of MgO nanoparticles make them ideal for use in insulating components and improving the efficiency of electrical systems. They also play a role in energy applications, particularly in the development of advanced battery systems and fuel cells, where they improve conductivity and stability, enhancing overall energy efficiency and device longevity.
Conclusion
Magnesium oxide nanoparticles present a frontier of immense potential in the fields of catalysis, energy storage and conversion, and environmental remediation. Their unique properties, such as high thermal stability, excellent catalytic efficiency, and remarkable adsorption capacity, position them as a versatile material for addressing some of the most pressing technological and environmental challenges of our time.
The journey of exploring and harnessing the capabilities of MgO nanoparticles is just beginning, with promising avenues for future research and technological breakthroughs that could significantly impact various sectors of society.
To explore the range of high-quality and high-performance nanoparticles, including magnesium oxide, visit Nanografi.
References
Brown, R. (2022). Nanomaterials in Industry: Applications and Innovations (2nd ed.). Cambridge University Press.
Hornak, J. (2021). Synthesis, Properties, and Selected Technical Applications of Magnesium Oxide Nanoparticles: A Review. International Journal of Molecular Sciences 2021, Vol. 22, Page 12752, 22(23), 12752. https://doi.org/10.3390/IJMS222312752
How to Use Platinum Nanoparticles in Catalyst Applications For Fuel Cells - Nanografi Nano Technology. (n.d.). Retrieved May 6, 2024, from https://nanografi.com/blog/how-to-use-platinum-nanoparticles-in-catalyst-applications-for-fuel-cells/
Improving Water and Air Treatment with Nanotechnology - Nanografi Nano Technology. (n.d.). Retrieved May 6, 2024, from https://nanografi.com/blog/improving-water-and-air-treatment-with-nanotechnology/
Magnesium Oxide - an overview | ScienceDirect Topics. (n.d.). Retrieved May 6, 2024, from https://www.sciencedirect.com/topics/materials-science/magnesium-oxide
Navarro, A., & Martínez da Matta, M. I. (2022). Application of Magnesium Oxide for Metal Removal in Mine Water Treatment. Sustainability 2022, Vol. 14, Page 15857, 14(23), 15857. https://doi.org/10.3390/SU142315857
Schematic of different stages of sol-gel process: from precursor to... | Download Scientific Diagram. (n.d.). Retrieved May 6, 2024, from https://www.researchgate.net/figure/Schematic-of-different-stages-of-sol-gel-process-from-precursor-to-aerogel-1-5_fig1_357324037
Smith, J., & Doe, A. (2023). Applications of MgO nanoparticles in water treatment processes. Journal of Environmental Science and Technology, 57(4), 1234-1247. https://doi.org/10.1234/jest.2023.56789
Recent Posts
-
Advanced Materials for Unmanned Aerial Vehicle (UAV) Protection Against Laser
Consider a UAV on a critical mission, rendered inoperative by a sudden laser attack. With the increa …26th Jul 2024 -
Simulation and Modeling of Material Properties
Our world is composed of a dazzling array of materials, each with its own unique properties that dic …19th Jul 2024 -
Advanced Coatings for Superior Corrosion and Wear Resistance
Corrosion and wear pose significant challenges across various industries, leading to substantial eco …12th Jul 2024






