2 Best Ways to Synthesis of Carbon Nanotubes
Carbon nanotubes are a nanoscopic configuration of carbon atoms arranged in the form of hexagonal meshes that form a cylindrical structure, similar to a tube.
The characteristics of these rolled graphene layer structures vary substantially depending on the orientation of the hexagons that form them about their axis, standing out because they surpass the most known materials in their physical properties. Due to their unique properties, they have a lot of important uses in different fields. This article aims to discuss the methods used to synthesize carbon nnotubes in detail. Explore Nanografi's various forms of carbon nanotubes that exhibit exceptional electrical and optical properties.
Introduction
In the field of nanomaterials, in the specific case of polymeric nanocomposites, new applications are opened by the use of carbon nanotubes as reinforcing agents as reinforcing agents dispersed in the continuous phase. These "nanotubes" can be considered both as reinforcing agents (relative to the mechanical properties) but also electrical and mechanical, which they can provide to the polymer matrix.
In fact, about their chemical-physical characteristics, carbon nanotubes show highly attractive properties. From the moment of their discovery 1991 as a waste product of the synthesis process of the parent molecule (fullerene) and the consequent in-depth study of their characteristics and potential applications, the interest on the part of researchers around the world has been gradually increasing.
One of the problems that has previously blocked a more invasive use of nanotubes in the most diverse fields is that of the inherent difficulties in synthesis processes at a sustainable price. In fact, currently on the market, it is only available in limited and very expensive quantities.
Another very important problem is the control of the size and morphology of nanotubes, ultimately making real nanotechnology. However, all this has led many research centers of various levels to undertake very in-depth studies and research on the subject. The potential applications are almost unlimited and range from polymeric nanocomposites with improved mechanical properties to all those applications where very special and peculiar electrical, thermal and optical properties are required.
Structure of Carbon Nanotubes
Carbon nanotubes were discovered accidentally by researcher Sumio Iijima, in the waste products in the fullerene production process. In fact, carbon nanotubes have a structure strongly linked to that of fullerene (third crystalline form of C60 carbon, the famous Buckyball very similar to a soccer ball). Carbon nanotubes are made up of flat graphite (graphene) sheets of carbon atoms to form a hexagonal structure rolled up to delineate a cylindrical structure.
Two forms of nanotubes are usually considered
- Single walled nanotubes (SWNT)
- Multi walled nanotubes (MWNT)
Single-walled carbon nanotubes have a diameter ranging from 1 to 10 nm, but generally the diameter is around 2 nm. Nanotubes are characterized by a high length / diameter ratio (10e4 10e5) and it is precisely for these and other characteristics that carbon nanotubes assume such captivating behaviors.
Between the walls of the multi-walled carbon nanotubes, weak interactions are established which seem to arise to stabilize the structure upon growth during the synthesis phase. The diameter of multi-walled nanotubes is generally much larger than single walled ones up to a few tens of nanometers.
There are also major imperfections of single-walled carbon nanotubes. For these reasons, single-walled nanotubes are much more valuable and very expensive commodities.
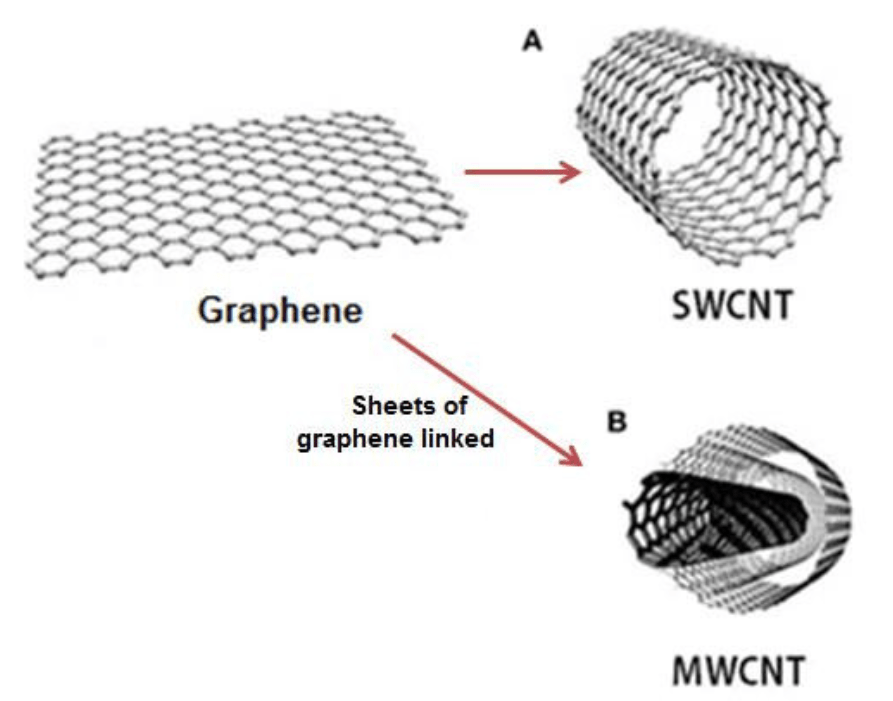
Figure 1. Graphene and carbon nanotubes as structures.
Production Methods of Carbon Nanotubes
There are two main methods to produce carbon nanotubes, high-temperature methods, and the Chemical Vapor Deposition method.
- High-Temperature Methods
- Chemical Vapor Deposition Method
1. High Temperature Method
This method used by Sumio Iijima consists of inserting two graphite rods, the anode and the cathode, in an electric arc production chamber, filled with non-reactive gases. When they are brought together and an electric current flows through them, an arc is produced, it is consumed to form a plasma whose temperature can reach 6000 °C, which instantly vaporizes the tip of one of the two rods. Carbon gas produced during this vaporization forms nanotubes which generally have 20 to 30 walls. If metal catalysts are added inside the anode, it is possible to obtain single-walled carbon nanotubes.
The synthesized nanotubes are almost perfect. However, the process is so complex that in the end, we have little control over the outcome. One of the main problems is the formation of other forms of carbon which are very difficult to separate from nanotubes, moreover, the high temperature necessary for the synthesis of nanotubes is restrictive for them, they tend to partially melt and adhere.
To get more information about the synthesis of carbon nanotubes, you can read our blog post here.
1.1 Laser ablation
Technique carried out by the group of R. Smalley, at the University of Houston (USA), which consists of bombarding a graphite rod with continuous laser radiation (with the continuous flow) or pulsed (with short pulses, it can be more powerful than the continuous laser). During this process, a water-cooled copper collector is strategically placed to capture the expelled carbon fragments. As the laser ablates the graphite, these fragments are rapidly cooled and deposited onto the collector.
Over time, this leads to the formation of a 'felt' of carbon nanotubes, which grows along the tip of the collector, benefiting from the quick thermal quenching provided by the cooling system. This not only enhances the yield but also improves the quality of the nanotubes formed. The graphite is either vaporized or expelled in small fragments of a few atoms, and the presence of the copper collector aids in the condensation of these carbon species into structured nanotubes. The process is more expensive, but has a higher synthesis yield than the electric arc production method; it also allows easier control, which allows the study of the synthesis conditions and their modeling to obtain only the desired products.
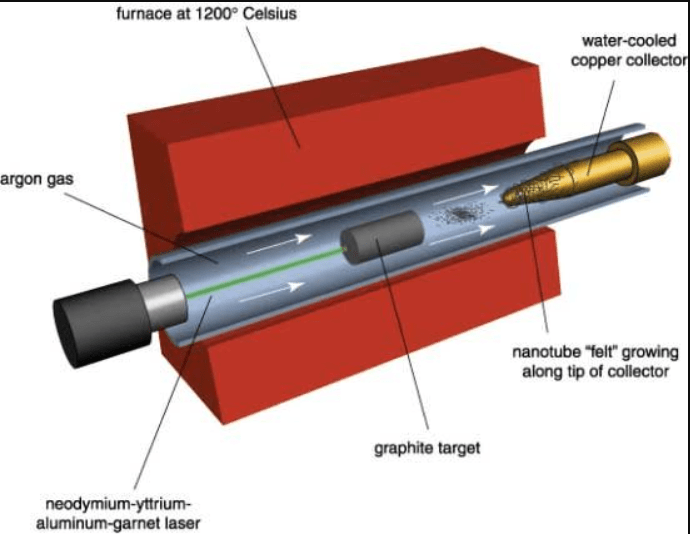
Figure 2. Laser ablation equipment to produce CNTs.
1.2 Solar Reactor
This method consists of concentrating solar energy on a graphite target to reach the vaporization temperature. This process makes it possible to synthesize an average of 0.1 g to 1 g of nanotube per "experiment", the production and operating mode is close to that by continuous laser, it is, therefore, suitable, like the latter, for in situ studies (behavioral study, carried out by observation in the situation).
Advantages and Disadvantages of High Temperature Methods
Advantages:
- Synthesis of single-walled nanotubes (whereas with the other methods one obtains only multi-walled nanotubes or an inseparable mixture)
- Formation of very pure products.
Disadvantages:
- Formation of clusters that must be dissociated to be able to apply them
- No control over the length of the nanotubes.
2. Chemical Vapor Deposition (CVD) Method
The CVD method is carried out using a liquid or gaseous carbon source to which a metallic precursor is added. Ferrocene, an organometallic compound, is frequently used, and sometimes nickelocene. The aerosol solution, which is transformed into fine droplets, is then transported by an inert gas, generally argon, to a furnace at a temperature between 750 °C and 900 °C.
In this environment, nanotubes "grow," either on the glass wall of the tube or on a silicon plate, placed to facilitate the recovery of the nanotubes. The continuous supply of reagents forces the nascent nanotubes to align vertically above the place where they "grow," taking up as little space as possible. As a result, multi-layered, aligned nanotubes with a length of approximately 200 micrometers are obtained. After the reaction, the nanotubes still contain some impurities, mainly the starting metal, which is iron or nickel. These impurities are removed by "re-baking" the nanotubes under an inert gas atmosphere, as the presence of dioxygen would destroy them. This "re-baking" also opens the half-fullerenes at the ends of the nanotubes, allowing impurities to come out and making the nanotubes even more rectilinear by eliminating any defects, such as parts of a "broken" graphene layer.
CVD from aerosols is a catalytic CVD method based on the catalytic decomposition of liquid or gaseous hydrocarbons, like toluene or acetylene, in the presence of a metallocene such as ferrocene. The specificity of this process is that it is carried out in a single step, with the reactor being supplied simultaneously with a catalytic source and a carbon source. Different equipment can be used depending on the direction of injection of the precursors and the size of the samples synthesized:
- equipment in vertical configuration for several hundred cm² samples,
- equipment in horizontal configuration for samples ranging from a few cm² to several hundred cm².
All this equipment is made up of three parts: an aerosol generator to produce droplets, often with an accompanying evaporator; a furnace housing a reactor; and a cooling and evacuation system.
To get more information about carbon nanotube sponges, you can read our blog post here.
Two types of aerosol generators are used: an ultrasonic aerosol generator or an injection system of the automotive injector type. The ultrasonic aerosol generator is composed of a piezoelectric ceramic which vibrates and creates, by micro-cavitation, an aerosol on the surface of the liquid, which is then transported by the gas flow. The injector system consists of a needle valve whose opening is controlled in duration and frequency and it operates in pulsed mode.
CVD for the Synthesis of Vertically Aligned Carbon Nanotubes and Graphene
The decomposition of the catalytic and carbon precursors occurs in two stages. Initially, the metallocene will decompose thermally giving rise to the formation of catalytic particles in the gas phase which will, in a second step, generate the catalytic decomposition of the carbon precursor. This results in the formation of a deposit on the walls of the reactor or various substrates (quartz, silicon, stainless steel, aluminum, carbon and carbon fiber fabrics, etc.). This deposit consists of carbon nanotubes aligned like a brush mat. Small equipment in the horizontal configuration is also used for the growth of graphene on metals.
Advantages and Disadvantages
- Formation of very pure products, practically flawless,
- Synthesis of multi-walled nanotubes,
- Formation of easily dissociable aligned nanotubes,
- High deposition temperatures generate residual stresses (material fatigue).
Properties of Carbon Nanotubes
Carbon nanotubes have some amazing qualities that make them stand out in science and engineering. They are incredibly strong and can withstand a lot of pressure without changing shape. At the same time, they are stiff and rigid on a tiny scale. Despite this rigidity, they're also flexible and can bend easily without snapping, which is useful for many applications that require both strength and the ability to flex.
These tiny tubes are also great conductors of electricity, allowing a lot of current to pass through a small area without a lot of resistance. In some special cases, they can even carry electrical currents without any resistance at all, which is known as superconductivity. This could lead to big changes in how electronic circuits and power systems are made.
In terms of heat, carbon nanotubes are very good at moving it away from where it's not wanted, which is important for managing temperatures in various devices. They also have a lot of surface area for their size, which makes them very reactive and excellent for speeding up chemical reactions, helping in catalysts, or acting as sensors. All these properties open up exciting possibilities in electronics, chemistry, and material sciences.
Applications of Carbon Nanotubes
Depending on the previous properties, the following potential or real applications arise
- Storage devices for fuel cells
- Reinforced Composite Polymers
- Conductive polymers
- Flame retardants
- Nanoelectronics
- Battery electrodes
- fuel Membranes
- Elements for interaction with light
- Solar cells
- Pharmaceutical transport and release systems
- X-ray devices
- Data storage (pc memories)
- Field emission display FED
- Sensors
- Artificial muscles
- Separation membranes.
- Chemical catalysis
Conclusion
Carbon nanotubes are similar to tiny rolled sheets of graphite with nanometric diameters and lengths on the order of microns. They are unique materials with exceptional mechanical, electrical, optical, thermal, and chemical properties that make them suitable for improving many existing products and even for generating new ones. It is manufactured by high temperature methods and chemical vapor deposition methods, each having its own advantages and disadvantages, as described above.
There are many applications that can obtain important benefits when incorporating carbon nanotubes. For example Composite materials reinforced with nanotubes, flat screens that use nanotubes as field emitters, biological and chemical sensors to detect pollutants, drug delivery, or fuel cells. In general, sectors such as electronics, materials, sensors, biotechnology, chemistry, energy, mechanics, scientific instrumentation, and photonics could be favored by the introduction of carbon nanotubes in many of their products.
As a leading supplier of high-quality nanomaterials and nanotechnology-based products, Nanografi always supports your projects and researches.
References
Applications of Carbon Nanotubes in Water Purification - Nanografi Nano Technology. (n.d.). Retrieved January 29, 2024, from https://nanografi.com/blog/applications-of-carbon-nanotubes-in-water-purification/
Applications of Multi-Walled Carbon Nanotubes - Nanografi Nano Technology. (n.d.). Retrieved January 29, 2024, from https://nanografi.com/blog/applications-of-multiwalled-carbon-nanotubes/
Carbon Nanotube (CNT) Sponges - Nanografi Nano Technology. (n.d.). Retrieved January 29, 2024, from https://nanografi.com/blog/carbon-nanotube-cnt-sponges/
Chemical vapor deposition - Wikipedia. (n.d.). Retrieved January 29, 2024, from https://en.wikipedia.org/wiki/Chemical_vapor_deposition
Chemical Vapor Deposition CVD Graphene - Nanografi Nano Technology. (n.d.). Retrieved January 29, 2024, from https://nanografi.com/blog/chemical-vapor-deposition-cvd-graphene/
Green Synthesis of Carbon Nanotubes - Nanografi Nano Technology. (n.d.). Retrieved January 29, 2024, from https://nanografi.com/blog/green-synthesis-of-carbon-nanotubes/
Laser ablation equipment to produce CNTs. Reprinted figure with... | Download Scientific Diagram. (n.d.). Retrieved January 29, 2024, from https://www.researchgate.net/figure/Laser-ablation-equipment-to-produce-CNTs-Reprinted-figure-with-permission-from-B_fig14_253505375
Nanotubes as Natural Means for Reinforcing Concrete - Nanografi Nano Technology. (n.d.). Retrieved January 29, 2024, from https://nanografi.com/blog/nanotubes-as-natural-means-for-reinforcing-concrete/
Recent Posts
-
Advanced Materials for Unmanned Aerial Vehicle (UAV) Protection Against Laser
Consider a UAV on a critical mission, rendered inoperative by a sudden laser attack. With the increa …26th Jul 2024 -
Simulation and Modeling of Material Properties
Our world is composed of a dazzling array of materials, each with its own unique properties that dic …19th Jul 2024 -
Advanced Coatings for Superior Corrosion and Wear Resistance
Corrosion and wear pose significant challenges across various industries, leading to substantial eco …12th Jul 2024








