What is the Difference Between Graphene Oxide and Reduced Graphene Oxide
Graphene oxide and reduced graphene oxide are valuable graphene derivatives. Graphene oxide and reduced graphene oxide show different chemical and structural properties due to the differences in their chemical compositions.
Introduction
The most prominent differences are observed in the electrical conductivity, hydrophilic behavior, mechanical strength, and dispersibility of these materials. Different properties of graphene oxide and reduced graphene oxide are utilized in energy storage applications, sensors, supercapacitors, solar cells, and biomedical applications.
Graphene and Its Derivatives
Allotropes of carbon such as diamond and graphite have been amongst the most important materials from the day of their discoveries. Graphite is composed of hexagonal carbon crystal sheets stacked on top of each other. It is a naturally occurring allotrope of carbon and the most stable form under standard conditions. Graphite has been used for several different purposes since the 1500s; however, most importantly, graphite has lead to the discovery of graphene.
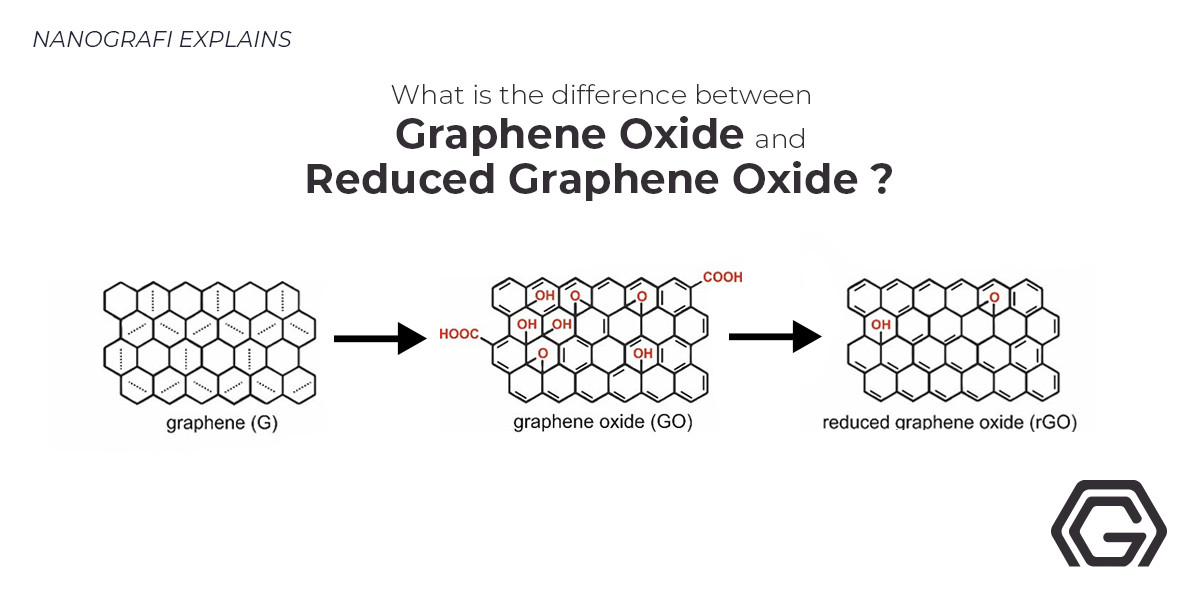
Graphene is defined as a one-atom-thick carbon sheet in a hexagonal lattice structure or as single-layered graphite. Frequently referred to as a “wonder” material, graphene has attracted a lot of attention due to its unique properties. It is the thinnest and lightest material known today. Its attractive properties include strong electrical conductivity (106 S cm−1), strong thermal conductivity (5000 W m−1 k−1), high mechanical strength (~40 N m−1), with Young's module of 1 TPa, optical transmittance (~97.7%) and large specific surface areas (~2600 m2 g−1). Unfortunately, the synthesis of this material is challenging especially on large scales. For this very reason, the discovery of graphene gave rise to its derivatives such as graphene oxide (GO) and reduced graphene oxide (rGO). The production of these materials is relatively easier on large scales.
Graphene Oxide
Graphene oxide is considered as the oxidized form of graphene. The discovery of graphene oxide goes way before the discovery of graphene. In 1859, GO was first synthesized by oxidation and exfoliation of graphite. However, until the discovery of graphene, graphene oxide remained rather insignificant. It was after the discovery of graphene that graphene oxide also attracted attention as a feasible way to obtain graphene. From that point on the studies on graphene oxide have picked up the pace and GO itself has found various different applications.
The synthesis of GO mainly achieved by the top-down approach which includes the treatment of graphite with strong oxidants or sulfuric acid and potassium permanganate, and the subsequent exfoliation step achieved by mechanical peeling methods such as sonication and shearing stress. However, is also possible to obtain GO through bottom-up synthesis methods such as chemical vapor deposition (CVD). Through the treatment process, the sp2 structure of graphite layers is disrupted and acquire several different oxygen-containing functional groups such as carboxyl, hydroxyl, or epoxy groups. The oxidation of graphite layers increases the interplanar spacing of the graphite structure. The subsequent exfoliation step separates the layers of graphite oxide to obtain a solution of homogenous graphene oxide layers. Different parameters such as initial oxidation conditions, energetic input, source, and lateral size of graphite used as a starting materialhave strong impacts on the resulted GO structure and the level of oxidation. Through the manipulation of these parameters, it is possible to obtain a variability in the properties of GO. Especially the level of oxidation strongly affects the chemical variability of GO sheets.
The disruption of the sp2 bonding network leads to distinctive properties. Graphene oxide shows low electrical conductivity causing an insulating or semi-conductive behavior depending on the degree of oxidation. The specific surface area of GO sheets is around 890 m2g-1. GO shows high mechanical strength with Young’s modulus of 207.6 ±23.4 GPa and fracture strength of ~120 MPa. The epoxy and hydroxy groups of GO sheets are located at the carbon basal plane while carboxy groups are located at the edges. The varying amounts of carbonyl, phenol, lactone, and quinine are also observed in the GO structure. The abundance of functional groups results in a hydrophilic behavior which is strongly dependant on the level of oxidation. GO sheets show good dispersibility as a result of their strongly charged nature and hydrophilicity. They form stable aqueous dispersions in a wide range of concentrations. Additionally, they are dispersible in organic solvents such as ethylene glycol, dimethylformamide (DMF), n-methyl-2-pyrrolidone (NMP), tetrahydrofuran (THF) due to the hydrogen bonding between the surface and solvent interface. Graphene oxide shows optical transparency stemming from the oxidized components while the graphene domains of GO show broadband fluorescence.
Reduced Graphene Oxide
The reduction of graphene oxide is considered to be an attractive route to obtain graphene-like behavior. Chemical, thermal, or photo-thermal reduction methods are used to obtain reduced graphene oxide structures. However, rGO cannot reach the pristine graphene structure. Even after severe reduction rGO still contains residual oxygen and structural defects originated in the chemical oxidation synthesis of GO.Chemical reducing agents used for rGO synthesis are often inorganic agents such as NaB or organics, such as phenyl hydrazine hydrate or hydroxylamine. Thermal reduction occurs in an inert or reducing atmosphere at temperatures between 300°C-2000°C.Finally, the photothermal-reduction of GO can be done with a direct laser beam at wavelengths under 390 nm (energy > 3.2 eV). The studies show that thermal reduction methods are advantageous compared to chemical reduction methods. Not only a lower level of reduction is achieved by chemical reduction methods but also the reducing agents used in methods are highly toxic. The thermal reduction methods have the upper hand due to their high level of reduction and relatively environmentally friendly process. The carbon to oxygen ratio of the resulting product is an important feature. The higher the C/O ratio the closer the properties of rGO to the properties of pristine graphene.
The reduction process causes drastic changes in the structural properties, mechanical strength, stability, dispersibility, and reactivity of GO. These changes are directly related to the elimination of the oxygen containing compounds in the GO structure and restoration of the sp2 structure after the reduction process. One of the most important effects of GO reduction process is the increase of electric conductivity up to 6300 S cm−1and high mobility of 320 cm2 V−1 s−1. The surface area of rGO also increases during the reduction process. rGO sheets show strong mechanical strength, with Young’s modulus of ~1.0 TPa and breaking strength of ~130 GPa which is similar to the graphene. As opposed to GO, reduced graphene oxide acquires a hydrophobic behavior due to the increased C/O ratio of the structure. Stemming from the hydrophobicity of rGO, the dispersibility of this material also decreases after reduction. In addition to the dispersibility, the colloidal behavior of rGO is also affected by the reduction process decreasing the critical coagulation concentration. Even though the graphene structure is not fully recovered by the reduction of GO. Reduced graphene oxide still holds useful properties such as controllable functionality, high electric and thermal conductance the availability of initial material, cheap and scalable preparation process.
To get more information about the uses of graphene in daily life,
you can read our blog post here.
Graphene Oxide vs. Reduced Graphene Oxide
Both graphene oxide and reduced graphene oxide have developed to be valuable derivatives of graphene. However, they show critical differences in both their structural and chemical properties. As it was mentioned in the above sections, the main difference between GO and rGO is the C/O ratio in their structure. While the C/O ratio is very low in GO structures, it is significantly higher in rGO structures approaching to almost zero oxygen content. The rest of the differences between GO and rGO materials mainly stem from this difference between C/O ratios. The most important difference is considered to be the electrical conductivity of these two materials. While GO show insulating or semi-conducting behavior, rGO shows high electrical conductivity 6300 S cm−1. The difference in the conductivities of GO and rGO opens up different application possibilities for these materials. Another significant difference between GO and rGO structures is the specific surface area. GO shows relatively lower surface area (890 m2g-1) in comparison to rGO structure which almost restores the extremely high surface area of pristine graphene (~2600 m2 g−1). The mechanical strength of GO is also found to be lower than the mechanical strength of the rGO structure. The Young’s modulus of GO is found to be almost half of rGO and graphene. Another important effect of the oxygen containing compounds is the hydrophilic behavior induced by the increased surface charge. Hence, GO structures show hydrophilic behavior while rGO shows hydrophobic behavior due to the loss of oxygen containing compounds. The differences between hydrophilic/hydrophobic behavior also induce a difference in the dispersibility of GO and rGO. Graphene oxide shows high dispersibility in aqueous media while rGO shows significantly lower dispersibility. For example, as the oxygen content of rGO is reduced from 31% to 9% the dispersibility of the material after sonication decreased from 8 to 2.5 µg/ml. Additionally, the colloidal behavior of the rGO follows the same trend as dispersibility. The different properties of GO and rGO have both found various applications in the scientific community and the industry.
Application Areas of Reduced Graphene Oxide and Graphene Oxide
Energy Storage
rGO is utilized in lithium-ion, lithium-sulfur, and lithium-oxygen batteries for energy storage applications. The high surface area of this material is a valuable advantage to obtain high capacity energy storage devices. Especially the highly conductive nature of rGO materials are utilized in the anode and cathode materials of these rechargeable batteries. The conductive carbon network promotes effective ion transfer and the exchange of electrons. Furthermore, the high diffusivity of Li on the graphene planes enhances the rate capability. For example, Fe2O3 nanoparticles decorated rGO platelets are used as an anode material for Li-ion batteries which exhibited discharge and charge capacities of 1693 and 1227 mAh g-1. Several different studies on rGO anode electrodes showed that rGO is a convenient material for the high capacity energy storage applications.
Also, free-standing rGO films are used as cathode material in Li-ion batteries successfully. On the contrary to anode materials, high content of functional oxygen containing compounds in the rGO structure is required for high capacity cathode electrodes.
Rechargeable batteries also utilize GO in their structure. Similar to rGO, the high specific surface area of GO material is important for the high capacity of these materials. Additionally, the oxygen containing compounds on the GO surface offer active bonding areas for electrochemical materials. Even though the electrical properties of GO can be tuned through changing the concentrations of oxygen containing compounds, the GO based anodes show poor cycling capacity due to the formation of solid electrolyte interphase (SEI) and the reaction of Li-ions with oxygen functional groups.
Although GO shows poor performance in anode materials of Li-ion batteries, they have been used in the cathode materials successfully. For example, the GO/LiFeSO4F composite serves as a cathode material for improved cycle stability and rate capability for lithium-ion batteries.
GO composites are also used in Lithium-sulfur batteries for the development of high energy density batteries. For example, Sulphur and lithium polysulphides can be immobilized on GO material by using the reactive functional groups on the GO structure. Strong interaction between GO and sulfur or polysulphides enables lithium/sulfur cells with a high reversible capacity of 950–1400 mAh g-1, and stable cycling for more than 50 deep cycles.
Solar Cells
The large specific surface area of GO and rGO is an extremely attractive property for solar cell applications. Due to its semiconducting properties GO is used as the hole transport and electron blocking layer and as an effective interfacial layer (IFL) in organic photovoltaics. GO significantly enhances the durability of devices by increasing the active layer-IFL interfacial stability under thermal and environmental stress.
Additionally GO is used in the cathode materials of dye-sensitized solar cells in combination with counter electrodes of rGO composites such as rGO–TaON composite and multiwalled carbon nanotube-rGO nanoribbon.
To get more information about the applications of graphene sheet films,
you can read our blog post here.
Sensors
Both GO and rGO are utilized in gas sensing applications. rGO attracts attention due to its high surface area and electrical conductivity while GO shows good sensing abilities due to active surface and high surface area. rGO/CuFe2O4 nanocomposite is used for a high–performance NH3 gas sensor which utilizes the conductivity of rGO and sensing ability of CuFe2O4. On the other hand, graphene oxide and cuprous oxide (GO/Cu2O) nanocomposite based sensors are used for trimethylamine (TMA) gas sensing. The system shows good sensitivity, reversibility, selectivity, and stability in 60 days.Additionally, both rGO and GO nanocomposites are found to be a good sensor for hydrogen, nitrogen dioxide, humidity.
Supercapacitors
Supercapacitors function based on electrochemical double-layer capacitance (EDLC) and release energy by nanoscopic charge separation at the electrochemical interface between an electrode and an electrolyte. rGO is considered a good candidate for new generation supercapacitor applications due to their high electrical conductivity, specific surface area, and cyclic stability. Several different rGO nanocomposites such as rGO/Zn/PCz, rGO-carbon black, and rGO/ZnO are utilized in the development of supercapacitor electrodes. The capacitance of these supercapacitors is enhanced up to 33.80 F/g resulting in high power (P = 442.5 W/kg) and energy-storage (E = 1.66 Wh/kg) capabilities. Additionally reduced graphene oxide is also used in the aerogel form for supercapacitor applications further increasing the surface area and consequently the capacitance of the supercapacitor.
However, compared to rGO, the studies on GO for supercapacitor applications are quite scarce due to the lower electrical conductivity of GO material.
Membranes
Chemically active nature and porous structure of graphene oxide have been utilized for the enhancement of membrane properties and separation performance. GO-polymer composites are employed for O2/N2 and CO2/N2 separation applications. Moreover, including GO into the membrane structure improves the mechanical properties of the membrane.
Biosensors
The composites of rGO and GO are used as optical, electrochemical, and field-effect transistor (FET) biosensors. These composites often contain metal nanoparticles such as platinum and silver or polymers. Biocompatible GO and rGO have been widely exploited for the sensing of biomolecules like glucose, DNA, D-glucosamine, microRNA, DNA/ RNA aptamers, multiplexed microRNA, optical aptamer, and arbitrary DNA mutations.
The fluorescent behavior of GO material is used for the optical biosensing applications to detect different biological molecules, such as cancer biomarkers glucose, H2O2, dopamine, NAs, food toxins, and metal ions.
Biomedical Applications
GO shows excellent DNA absorption properties and biocompatibility. DNA binding to GO is found to be very stable and reversible. These properties make it possible to prepare DNA-based graphene materials for various bio-applications. GO-based materials especially attract attention in drug delivery. GO nanosheets show very low cytotoxicity and high cellular uptake hence, explored as the ideal nanocarriers for drug delivery and intracellular fluorescent nanoprobe. Various graphene-based composites like patterned substrates of nano-GO, GO/hydrogel-based angiogenic, hyaluronic acid-decorated GO nanohybrids, functionalized GO nanoparticles, and fluorescent GO via polymer grafting were utilized for efficient drug delivery.
In addition to drug delivery applications, GO-based materials are also used for diagnostic and photothermal therapy applications. Fluorogenic resveratrol-confined GO and hybrid GO based plasmonic-magnetic multifunctional nanoplatforms are utilized for diagnosing Alzheimer's disease. In the sense of therapeutic applications, the combination of gold nanostars and GO were utilized for ultraefficient photothermal cancer therapy. Additionally, excellent photothermal effect confers rGO the great potentials in photothermal therapy against cancer and heat-induced controlled drug release.
Finally, both rGO and GO shows promising antibacterial properties with a broad antibacterial spectrum due to their special physicochemical properties and unique antibacterial mechanism. Metals such as silver and gold are often used as nanocomposites materials with GO and rGO to enhance the antibacterial activity of these structures. ZnO/GO composites of varying contents of ZnO in high quality and these composites own superior antibacterial properties against E. coli with low cytotoxicity. The antibacterial properties of GO based materials are also utilized to kill dental pathogens.
Conclusion
Graphene and graphene derivatives have become important materials because of their unique properties. Especially, graphene oxide and reduced graphene oxide are considered to be valuable, low-cost, easily attainable derivatives of graphene. rGO attracts attention due to its similar properties to pristine graphene. GO has first attracted attention as an intermediate material for graphene synthesis however, since then it has been utilized for various different applications. GO is obtained by oxidation and exfoliation of graphite layers and contains a considerable amount of oxygen containing compounds such as carboxyl, hydroxyl or epoxy groups on its carbon basal plane and edges. rGO is obtained by chemical or thermal reduction of GO causing a significant decrease in the amount of oxygen containing groups. As the C/O ratio increases rGO structure and properties become more and more similar to pristine graphene.
Due to the chemical and structural differences, GO and rGO show different mechanical, electrical, and chemical properties. One of the most important differences between GO and rGO is the electrical conductivity of these materials. While GO shows insulating or semi-conducting behavior rGO shows excellent electrical conductivity which is almost as good as pristine graphene. The elimination of oxygen containing compounds leads to a higher specific surface area for rGO materials compared to GO. Another distinctive property is hydrophilic/hydrophobic behavior. GO shows strong hydrophilic behavior while rGO shows hydrophobic behavior. Moreover, the mechanical properties of rGO are far superior to the mechanical properties of GO. On the other hand, GO shows much better dispersibility and colloidal properties compared to rGO.
Interesting properties of GO and rGO are utilized in various different applications. Nanocomposites of GO and rGO are commonly used in energy storage applications, sensors, biosensors, biomedical applications, solar cells, and supercapacitors. The high surface area of both materials is a major advantage for all of these application areas. The high conductivity of rGO is utilized to improve the capacity and cyclic stability of the Li-ion batteries’ anode material. On the other hand, the chemical activity of GO is utilized in the cathode of Li-ion batteries. In addition to Li-ion batteries GO and rGO are also utilized for high capacity applications in Lithium-sulfur and lithium-oxygen batteries. Similarly, GO and rGO are used to increase the capacitance of supercapacitors. Biocompatibility and DNA binding properties of GO are found to be useful in biomedical applications such as drug delivery, diagnostics, and therapeutic applications. GO-based nanocomposites are used for cancer-treatment applications and diagnosing Alzheimer’s disease. All in all, graphene derivatives GO and rGO are promising materials for graphene-based applications in energy storage, electronics, sensors, and bio-based applications.
To get more information, you can visit Blografi.
References
Raslan, A., del Burgo, L. S., Ciriza, J., & Pedraz, J. L. (2020). Graphene oxide and reduced graphene oxide-based scaffolds in regenerative medicine. International Journal of Pharmaceutics, 119226.
Suk, J. W., Piner, R. D., An, J., & Ruoff, R. S. (2010). Mechanical properties of monolayer graphene oxide. ACS nano, 4(11), 6557-6564.
Ling, S. (2019). Structure and synthesis of graphene oxide. Chinese Journal of Chemical Engineering.
McCoy, T. M., Turpin, G., Teo, B. M., & Tabor, R. F. (2019). Graphene Oxide: Surfactant or Particle?. Current opinion in colloid & interface science.
Lawal, A. T. (2019). Graphene-based nano composites and their applications. A review. Biosensors and Bioelectronics, 111384.
Singh, R. K., Kumar, R., & Singh, D. P. (2016). Graphene oxide: strategies for synthesis, reduction and frontier applications. RSC advances, 6(69), 64993-65011.
Xia, M. Y., Xie, Y., Yu, C. H., Chen, G. Y., Li, Y. H., Zhang, T., & Peng, Q. (2019). Graphene-based nanomaterials: the promising active agents for antibiotics-independent antibacterial applications. Journal of Controlled Release.
Recent Posts
-
Advanced Materials for Unmanned Aerial Vehicle (UAV) Protection Against Laser
Consider a UAV on a critical mission, rendered inoperative by a sudden laser attack. With the increa …26th Jul 2024 -
Simulation and Modeling of Material Properties
Our world is composed of a dazzling array of materials, each with its own unique properties that dic …19th Jul 2024 -
Advanced Coatings for Superior Corrosion and Wear Resistance
Corrosion and wear pose significant challenges across various industries, leading to substantial eco …12th Jul 2024