Graphene Sheet Uses
In recent years, one of the most significant discoveries in the world of nanotechnology, graphene, is a single layer of carbon atoms and holds the potential to revolutionize a myriad of fields from electronics and energy storage to medicine and environmental science.
Throughout this article, we will embark on a comprehensive exploration of the multifaceted uses of graphene sheets, delving deep into its applications that span from next-generation electronics, sophisticated energy storage solutions, and cutting-edge medical advancements, to its role in addressing some of the most pressing environmental challenges of our time. Meet our high-performance and high-quality products with Nanografi, one of the world's leading graphene producers.
Introduction
Graphene is a remarkable material composed of a single layer of carbon atoms arranged in a hexagonal lattice. It can be visualized as a two-dimensional sheet or a single atomic layer of graphite. Although technically a non-metal, graphene exhibits properties similar to those of a semi-conducting metal, earning it the nickname "quasi-metal." These unique properties stm from the sp2 hybridized C-C bonds and the π-electron clouds that hover above and below the sheet. What makes graphene especially significant for engineers and technologists is its combination of intriguing structural and physical characteristics. Even thin flakes consisting of just a few layers of carbon atoms, including monolayer graphene, have garnered immense interest due to their potential for a wide range of technological applications.
Overview of Graphene
Graphene has gained enormous interest amongst scientists since its discovery due to its excellent properties. Graphene was first isolated in 2004 by two researchers, Andre Geim and Kostya Novoselov, at the University of Manchester. They won the Nobel Prize in 2010 in Physics for their revolutionary work. Graphene is a one-atom-thick layer of sp2-hybridized carbons atoms tightly arranged into a 2D honeycomb lattice.
Carbon is one of the most abundant elements inside the human body. Moreover, it is also one of the most abundant elements by mass in the universe. So, all living materials have a chemical basis made up of carbon. So, graphene can be an ecologically friendly material for numerous applications. The band structure of graphene is unique. The valence and the conduction bands touch each other at six Dirac points exhibiting zero band gap semiconductor behavior. It has high fracture strength, excellent electrical and thermal conductivity, fast mobility of charge carriers (2 × 105 cm2/Vs, 200 times higher than silicon), large specific surface area (2600 m2.g-1) and biocompatibility. All these properties are not only exceptional but also superior to other materials.
Carbon has a unique feature of forming a chemically stable two-dimensional one atom thick sheet called as graphene. Graphene is about 300 times stronger than steel. It is the lightest material known so far. One square meter weighs about 0.77 mg. It has electron mobility more than 15,000 cm2 V-1 s-1. It has tensile stiffness to the tune of 150,000,000 psi. It is the best conductor of heat at room temperature. Graphene is reported to have an exceptionally high value of thermal conductivity of approximately 5300 Wm-1 K-1. It has low coefficient of thermal expansion and high optical transmittance.
Graphene, due to its exceptional chemical and physical properties, has attracted attention in a wide range of application areas. Due to the remarkable electronic properties of graphene such as high charge transport mobility, it is a key material particularly in the manufacture of sensitive nanodevices, nanoelectronics, and nanosensors. Further, graphene is a promising building block for high performance electronics, sensors, and energy storage devices. Graphene nanostructures are integrated into a variety of electronic and optoelectronic applications, such as gas sensors, transistors, solar cells, and liquid crystal elements, due to its low electrical noise, and ballistic transport properties.
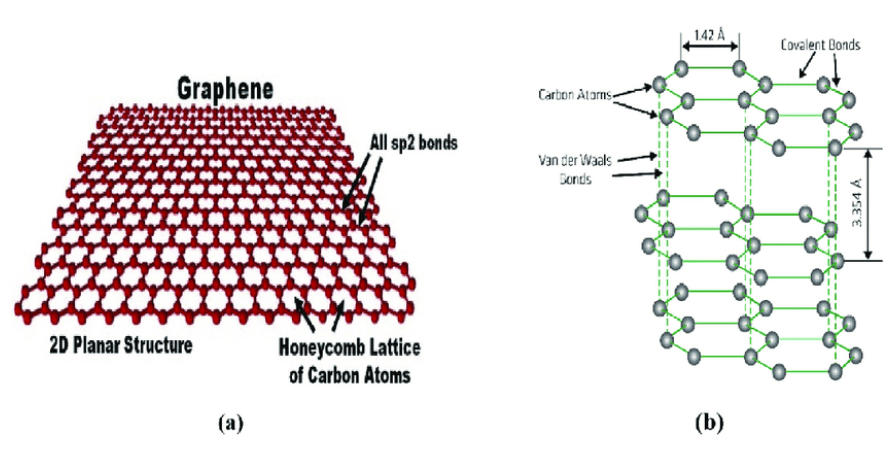
Figure 1. (a) Carbon atoms closely bonded in a honeycomb lattice with sp2 hybridization, forming a tightly packed arrangement, and (b) The atomic structure of graphene.
What is Graphene Sheet?
Graphene is one of the thinnest materials known to man, comprising a single layer of carbon atoms densely arranged in a hexagonal pattern. Despite being just one atom thick, these sheets are renowned for their impressive electrical conductivity at room temperature. Often described as the world's slimmest material, a graphene sheet extends from nanoscale dimensions to several microns, with every carbon atom covalently bonded to three others in a hexagonal arrangement. Recognizing these sheets usually involves microscopic imaging, which is crucial in ensuring quality during production. This immediate feedback helps refine the manufacturing process. Moreover, graphene's unique properties make it an excellent filler to boost electrical and mechanical features in composite materials.
Applications of Graphene Sheet
Graphene Batteries
Graphene enhanced Li-ion batteries show incredible characteristics such as longer lifespan, higher capacity, and faster charging time as well as flexibility and lightness, so that it could be used in wearable electronics.
To learn about graphene batteries, how they work, and how they differ from other batteries, read our blog.
Thermoelectric Devices
Seebeck effect is defined as a thermoelectric effect occurring when heat is applied to one of the two dissimilar electric conductors (or semiconductors) to move the electrons from the hot part to the cooler part and produce electricity. However, the energy generated by this method is really small, usually quantified by microvolts. Still, it is believed that it can be used to benefit from the heat generated by the engines, which is practically wasted. Graphene can be used to increase the Seebeck effect created by Strontium Titanate, almost up to 5 times.
Graphene Sheet in Nuclear Plants
Heavy water used in nuclear power plants to cool the reactors is both costly to produce and causes a million tons of CO2 emissions during production. Researchers from University of Manchester have discovered that there is a greener and low-cost method to produce heavy water: graphene membranes.
Graphene Sheet in Fuel Cells
It is known fact that even hydrogen atoms, known as the smallest atom, cannot pass through graphene. In a research, Sir Andre Geim and his team have tested if protons would be blocked by graphene or not. Suprisingly, protons could pass through graphene. This property would improve fuel cells performance by lowering the fuel crossover which is a major problem with fuel cells that decreases durability and efficiency.
To learn about the fuel cell development with nanotechnology, read our blog.
Drug Delivery & Cancer Treatment
Recently graphene has showed promise similar to carbon nanotubes in various biomedical applications such as drug delivery and cancer therapy. Graphene can provide a larger specific surface area than other commonly used carbon nanomaterials and forms strong pi bonds with the drug molecules which can therefore act as a good candidate for drug loading.
Graphene UV Sensors
UV sensors are used for detecting dangerous levels of ultra-violet radiation which can lead to skin problems or even cancer. However, it is not the only use of UV sensors, they are used in the military, optical communication, and environmental monitoring as well. On its own, graphene may not present a high photoresponsivity but when it is combined with other materials, they create flexible, transparent, environmentally-friendly and low-cost UV sensors which will lead to technologies such as wearable electronics in the close future.
Touchscreens
Indium tin oxide (ITO) is the commercial product used as transparent conductor of the smartphones, tablets, and computers. Researchers from the Rice University have developed a graphene-based thin film to be used in touchscreens. It is found that graphene-based thin film beats ITO and any other materials in terms of performance because it has lower resistance and higher transparency. Thus, graphene is the new candidate material for the replacement of ITO. Do you know what other electronic fields graphene is used in? Read our blog to find out.
Hard Drives and Memories
Usually, graphene is not considered magnetic, at least not in a controllable or useful way. In 2015, researchers from U.S. Naval Research Laboratory have found a way to turn graphene into a reliable and controllable electromagnetic material. If this innovation is used in hard drives, it is expected to have a capacity almost a million times greater than what we use today.
Superconductors
Scientists have discovered that graphene can also be used as a superconductive material. Two layers of Graphene can conduct the electron without any resistance. This can be accomplished by twisting these two layers of graphene at a ‘magic angle’ which is 1.1°. Most of the superconductive materials show their properties at temperatures close to absolute zero. Even High temperature superconductive materials relative to usual ones can work at around -140°C. In other words, these superconductive materials require a huge energy for cooling. If graphene can be used as a superconductive material at temperatures close to room temperature, there will be a huge revolution for many application areas.
Optoelectronics
A research conducted by the collaboration of different universities has shown that integrating graphene with silicon can beat current silicon photonic technology because devices made by graphene are cheaper, simpler and work at high-scale wavelengths. Apparently, graphene will present a low-energy optical telecommunication and many other convenient optical systems.
Food Packaging
Graphene can also be used as a coating material because it prevents the transfer of water and oxygen. Graphene sheets can be used in food or pharmaceutical packaging by keeping food and medicines fresh for longer time. It may seem a simple application, but it can dramatically reduce the amount of food waste people throw away every day.
To discover about all the uses and applications of graphene, you can read our most popular blog.
Graphene Sheet in Water Purification
Normally, water purification is not a simple process and feasibility of the process depends on how heavily the water is contaminated. An Australian scientist has found a low-cost technique to purify water at one step. Soybean-based graphene sheet, which is also called ‘GrapHair’, is used as a filter. This filter can make dirty water drinkable. it is more efficient, cheaper and environmentally friendly compared to other methods.
If you are interested in the use of graphene in water filtration, you can visit our blog page.
Helmets
An ideal helmet would be strong, resistant to impact, durable, comfortable, and light. Graphene is incredibly strong, light, and flexible. It’s even used in bulletproof vests, so it can resist impacts. With these properties, graphene is commercially used in motorbike helmets.
Insulation Material
Graphene can be used as a superconductor or insulator material when two sheets of graphene are arranged at a magic angle. Most of the metal parts of the cars, ships or planes suffer from rusting. When graphene is combined with paint, it can be a great insulation material for creating rust-free surfaces. Another application can be coating of bricks and stones. In this way, water-proof houses can be constructed.
Automotive
The extraordinary strength and hardness of graphene, coupled with its flexibility, is perfect to start creating cars that are immune to shocks. Moreover, accident-proof vehicles could also be created. This would result in a direct decline in road mortality. Graphene cars, which we may see in the showrooms within a decade, are also expected to be cheaper and lighter.
Radiation Shielding
Scientists have been trying to keep the radiation at minimum since it is very dangerous for human health. For this purpose, a variety of materials can be used as a shielding material to radiation but there are many parameters that affects the efficiency of shielding. Graphene is known as a weak radiation absorber, but scientists have found that it can be a great shielding material when it is used in multi-layered form which are graphene slabs. Graphene is an outstanding material for this purpose thanks to its low manufcturing cost, light weight and high efficiency compared to any other shielding materials.
Thermal and Infrared Vision
A great advance that we could see is the development of graphene lenses that allows thermal and infrared vision. The graphene allows manufacturing such ultrathin devices with a built-in camera that gives the user an infrared and thermal vision. Something that until now we have only seen in science fiction movies.
Conclusion
Graphene, due to its exceptional chemical and physical properties, has attracted attention in a wide range of application areas. Due to the remarkable electronic properties of graphene such as high charge transport mobility, it is a key material particularly in the manufacture of sensitive nanodevices, nanoelectronics, and nanosensors. Further, graphene is a promising building block for high performance electronics, sensors, and energy storage devices.
To get more information, you can visit Blografi.
References
Avouris, P., 2010. Graphene: Electronics and photonics properties and devices. Nano Letters 10, 4285–4294
Chakraborty, M., & Hashmi, M. S. (2018). Graphene as a Material – An Overview of Its Properties and Characteristics and Development Potential for Practical Applications. Reference Module in Materials Science and Materials Engineering. doi:10.1016/b978-0-12-803581-8.10319-4
Chung, C., Kim, Y.K., Shin, D., et al., 2013. Biomedical applications of graphene and graphene oxide. Accounts of Chemical Research 46 (10), 2211–2224
Graphene Batteries - Nanografi Nano Technology. (n.d.). Retrieved February 7, 2024, from https://nanografi.com/blog/graphene-batteries/
Graphene structure showing: (a) sp 2 hybridized closely packed carbon... | Download Scientific Diagram. (n.d.). Retrieved February 7, 2024, from https://www.researchgate.net/figure/Graphene-structure-showing-a-sp-2-hybridized-closely-packed-carbon-atoms-tightly_fig4_337003782
Military Applications of Graphene - Nanografi Nano Technology. (n.d.). Retrieved February 7, 2024, from https://nanografi.com/blog/military-applications-of-graphene/
Penkov, O. V. (2020). Preparation and characterization of graphene. Tribology of Graphene, 51-90. doi:10.1016/b978-0-12-818641-1.00003-4
Premkumar, T., Geckeler, K.E., 2012. Graphene–DNA hybrid materials: Assembly, applications and prospects. Progress in Polymer Science 37, 515–529.
Sakhaee-Pour, A., Ahmadian, M.T., Vafai, A., 2008. Potential application of single-layered graphene sheet as strain sensor. Solid State Communications 147, 336–340.
Shao, Y., Wang, J., Wu, H., et al., 2010. Graphene based electrochemical sensors and biosensors: A review. Electroanalysis 22, 1027–1036
Use of Graphene in Water Filtration - Nanografi Nano Technology. (n.d.). Retrieved February 7, 2024, from https://nanografi.com/blog/use-of-graphene-in-water-filtration/
What is Graphene: The Ultimate Guide - Nanografi Nano Technology. (n.d.). Retrieved February 7, 2024, from https://nanografi.com/blog/what-is-graphene-the-ultimate-guide/
Zhang, B., Wang, Y., Zhai, G., 2016. Biomedical applications of the graphene-based materials. Materials Science and Engineering C 61, 953–964.
Recent Posts
-
Advanced Materials for Unmanned Aerial Vehicle (UAV) Protection Against Laser
Consider a UAV on a critical mission, rendered inoperative by a sudden laser attack. With the increa …26th Jul 2024 -
Simulation and Modeling of Material Properties
Our world is composed of a dazzling array of materials, each with its own unique properties that dic …19th Jul 2024 -
Advanced Coatings for Superior Corrosion and Wear Resistance
Corrosion and wear pose significant challenges across various industries, leading to substantial eco …12th Jul 2024







