What Is LK-99: This Material Could Change Everything - Nanografi
What is the LK-99?
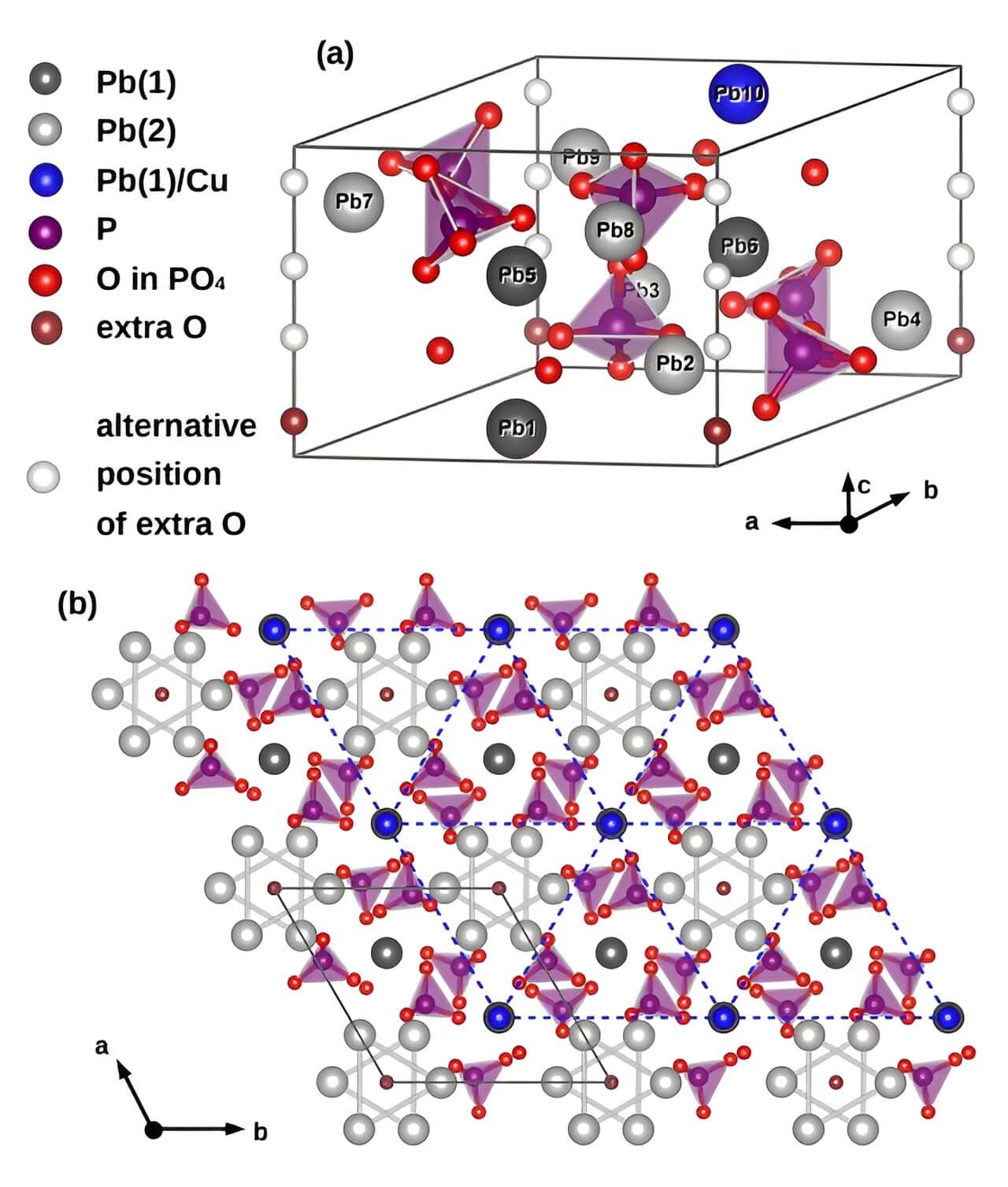
LK-99, a gray-black polycrystalline compound with a hexagonal crystalline pattern, was first discovered in 1999 by Lee Sukbae and Kim Ji-Hoon from South Korea University. This copper-enriched lead-oxyapatite substance is believed to function as a room temperature superconductor, allowing it to transmit electricity with zero resistance.
In the world of science, there are some differences of opinion regarding whether LK-99 exhibits superconductivity at room temperature. One group of scientists claims that LK-99 demonstrates superconducting properties at room temperature and standard atmospheric pressure, while other scientists are of the opinion that further research is necessary to evaluate the potential applications.

What are the components of LK-99?
The chemical composition of LK-99 is approximately Pb9Cu(PO4)6O. In this formula:
- Pb is lead
- Cu is copper
- P is phosphorus
- O is oxygen
The copper atoms are randomly distributed in the lead‒oxyapatite lattice. The doping of copper into lead‒oxyapatite is thought to be responsible for the room-temperature superconductivity of LK-99.
How to synthesize LK-99?
Lee and his team present a method for the chemical synthesis of LK-99 in three steps. Firstly, they produce lanarkite by mixing lead(II) oxide (PbO) and lead(II) sulfate (Pb(SO4)) powders in a 1:1 molar ratio and heating at 725°C for 24 hours:
PbO + Pb(SO4) → Pb2(SO4)O.
In the next step, they produce copper(I) phosphide (Cu3P) by mixing copper (Cu) and phosphorus (P) powders in a 3:1 molar ratio in a sealed tube under vacuum and heating to 550°C for 48 hours:
3 Cu + P → Cu3P.
Subsequently, the lanarkite and copper phosphide crystals are ground into a powder, placed in a sealed tube under vacuum, and heated to 925°C for between 5 to 20 hours:
Pb2(SO4)O + Cu3P → Pb10-xCux(PO4)6O + S (g), where 0.9 < x < 1.1.
There were several issues with this initial synthesis. The reaction was not balanced, and other researchers also reported the presence of copper(I) sulfide (Cu2S). The proposed balanced reaction is:
5 Pb2SO4O + 6 Cu3P → Pb9Cu(PO4)6O + 5 Cu2S + Pb + 7 Cu.
Many synthesis produced fragmentary results in different phases. Some fragments were responsive to magnetic fields while others were not. The first synthesis that produced pure crystals found these crystals to be diamagnetic insulators.
LK-99 Materials
Copper (Cu)
Copper is one of the main components of LK-99. It is a dopant, which means that it is added to the other elements in LK-99 to change its properties. In the case of LK-99, copper doping is thought to increase the conductivity of the material, which makes it a potential superconductor.
More detailed explanation of the role of copper in LK-99:
- Copper is a metal with high electrical conductivity. This means that it can carry electricity with very little resistance.
- When copper is added to LK-99, it disrupts the crystal structure of the material. This can create new energy levels for electrons, which can make it easier for them to flow through the material without resistance.
- This increased conductivity is thought to be the reason why LK-99 is a potential superconductor. However, more research is needed to confirm this.
It is important to note that the claim that LK-99 is a room-temperature superconductor has been disputed by other scientists.
Phosphorus (P)
More research is needed to fully understand the role of phosphorus in LK-99. However, the fact that phosphorus is a key component of this material suggests that it plays an important role in its superconducting properties.
Some explanations for the role of phosphorus in LK-99:
- Phosphorus acts as a dopant. Dopants are impurities that are added to a material to change its properties. In the case of LK-99, phosphorus may act as a dopant, disrupting the crystal structure of the material and creating new energy levels for electrons. This could make it easier for electrons to flow through the material without resistance, leading to superconductivity.
- Phosphorus is essential for the formation of the crystal structure of LK-99. The specific crystal structure of LK-99 is not fully understood, but it is thought to be important for its superconducting properties. Phosphorus may play a role in stabilizing this crystal structure, making it possible for LK-99 to superconduct at room temperature.
- Phosphorus interacts with the other elements in LK-99 to create a new state of matter. Superconductivity is a state of matter in which a material conducts electricity with zero resistance. It is possible that phosphorus interacts with the other elements in LK-99 to create a new state of matter that exhibits superconductivity at room temperature.
Lead Oxide (PbO)
- Lead oxide is an inorganic compound with the chemical formula PbO. It is a white, odorless, and tasteless solid that is insoluble in water. Lead oxide is a relatively inert substance that is used in a variety of applications, including lead-based paint, ceramics, and batteries.
- Lead oxide is produced by heating lead metal in air. It can also be produced by the reaction of lead carbonate or lead nitrate with nitric acid. Lead oxide is a strong oxidizing agent and can react with other metals to form lead alloys.
In the synthesis of LK-99, an amorphous precursor is used that is obtained from lead oxide apatite (Pb5(PO4)3OH) using lead oxide (PbO) and phosphoric acid (H3PO4). This precursor is then subjected to a series of heat treatments and eventually converted to LK-99.
Why is everyone talking about LK-99?
The main reasons for everyone talking about LK-99 include being a new material with the potential to revolutionize various industries. LK-99 stands out as the first material to exhibit superconductivity at room temperature. Room-temperature superconductivity is the ability to conduct electricity without any resistance at room temperature. This could enable the development of extremely efficient and powerful electric motors, generators, and transformers. It could also be used to create new types of sensors and medical devices.
Why room-temperature superconductors are so important?
Room-temperature superconductors are important because they can conduct electricity without resistance at temperatures achievable with everyday cooling methods. This has numerous practical implications:
- Energy Efficiency: They could drastically reduce energy loss during electricity transmission, making power grids more efficient and reducing costs.
- Renewable Energy: They could improve energy storage and transmission for renewable sources, increasing their viability and impact.
- Electronics: Room-temperature superconductors could lead to smaller, faster, and more energy-efficient electronics.
- Medical Imaging: Medical devices like MRI machines could become more affordable and accessible.
- Transportation: They could revolutionize high-speed transportation and make it more energy-efficient.
- Scientific Research: They could enable stronger magnetic fields for scientific research.
- Industrial Applications: Industries could benefit from improved energy efficiency.
- Environmental Impact: Reduced energy waste could lower greenhouse gas emissions.
While challenging, achieving room-temperature superconductivity would bring significant advancements across various sectors.
What are the future application areas for LK-99?
LK-99 is considered a breakthrough that could have far-reaching implications. Despite still being in the developmental stage, LK-99 holds the promise of being a major game-changer in the future.
Energy Sector:
-Enhanced electricity generation and distribution efficiency.
-More efficient electric motors, generators, and transformers.
-Potential reduction in energy consumption and environmental impact.
Electronics and Technology:
-Development of high-performance electronic components with minimal energy loss.
-Faster and more efficient electronic devices.
Transportation:
-Advancements in magnetic levitation technology for high-speed trains and vehicles.
-Creation of innovative propulsion systems for space exploration.
Medicine and Healthcare:
-Improved medical imaging devices and diagnostics.
-Enhanced capabilities for magnetic resonance imaging (MRI) machines.
-Development of more effective medical sensors and treatment devices.
Sensors and Detection:
-Creation of highly sensitive sensors for detecting various substances and environmental changes.
-Applications in pollution monitoring, security systems, and quality control.
Scientific Research:
-Improved capabilities for studying and manipulating materials at extreme conditions.
-Advancements in quantum research and understanding of fundamental physics.
Industrial Applications:
-More efficient industrial processes through improved power transmission and utilization.
Environmental Monitoring:
-Monitoring and management of environmental parameters with high precision.
Communication:
-Enhanced signal transmission in communication systems.
Renewable Energy:
-Improved efficiency and performance of renewable energy systems.
Aerospace:
-Development of new propulsion systems for aircraft and spacecraft.
References
https://nanografi.com/lk-99-raw-materials/
https://en.wikipedia.org/wiki/LK-99
https://cybernews.com/editorial/the-lk-99-controve...
https://phys.org/news/2023-08-newly-material-prope...
Recent Posts
-
MXenes from MAX Phases
MXenes, a group of two-dimensional materials derived from MAX phases, are gaining significant tracti …19th Oct 2024 -
Preserving History with Graphene's Power
Cultural artifacts are at risk of deterioration over time due to the destructive effects of both na …11th Oct 2024 -
The Role of Graphene in Neuroelectronics
Neuroelectronics is an interdisciplinary field that aims to develop devices that can interact with t …4th Oct 2024





