Lithium-Graphite Paste: An Article Review
Lithium-Ion batteries have been a significant invention due to their high energy density. Many researchers are trying to improve lithium-ion batteries since they are used in portable electronic devices and electric vehicles which are required to have long battery life in these days. High energy density, fast rate response, low cost and high safety are the most important parameters for an ideal lithium-ion battery. These parameters are achieved by maximizing the energy density by highcapacity cathodes, silicon-based anodes and organic liquid electrolytes. However, the problem is that operation of lithium-ion batteries is exothermic and organic liquid electrolytes are highly flammable which cause a huge heat generation. This problem affects the safety parameter negatively. For this reason, solutions for safety problems are required. Here we will review a solution brought in by Chinese scientists in their article “Lithium-Graphite Paste: An Interface Compatible Anode for Solid State Batteries” published in the journal “Advanced Materials”.
Instead of using organic liquid electrolytes, inorganic ceramic solid-state electrolytes (SSE) have been started to be used as an alternative way for the All-solid-state batteries (ASSBs) because SSE is non-flammable and it has enough toughness to prevent the internal short circuit from lithium dendrite. Therefore, it has been demonstrated that inorganic ceramic solid-state electrolytes exhibit a great safety level under rigorous conditions.
In this study, it was shown that garnet-type oxides are the most stable SSEs with lithium metal anode. However, it was also seen that lithium/garnet interface has a high impedance due to poor interfacial contact. As a result of this problem, many studies have been conducted to coat the garnet with different materials such as metal, metal oxides, semi-conductors, polymer interlayers and graphite for making garnet lithophilic. Researchers, from Tongji University, found a method to synthesize a ceramic compatible lithium anode by using graphite additives.
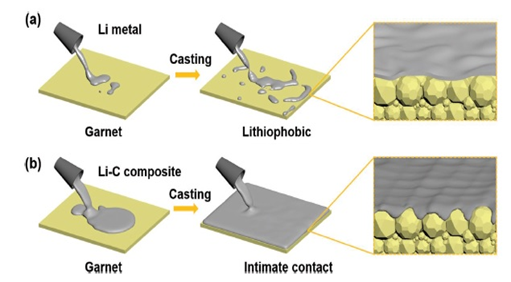
Figure 1: Schematic diagrams of casting pure lithium (Li) and lithium-graphite composite on garnet SSEs.
As can be seen from the Figure 1, poor contact is observed between garnet and pure lithium and Li-C composite can spread well on the garnet giving a perfect contact. Li-C composite shows lower fluidity and higher viscosity compared to pure Li. While pure Li/garnet interface delivers an interfacial resistance of 381 Ω cm2, Li-C/garnet interface delivers an interfacial resistance of 11 Ω cm2 which is remarkably low compared to pure Li. Thus, it can be said that Li-C cells outperforms the cells with pure Li.
Li-C composite is prepared by adding graphite powders in molten Li with continuous stirring and heating at 250°C. In addition, since graphite is abundant and relatively cheap, it offers lower cost and it does not need any complicated equipment and surface treatment. Thus, Li-C composite anode is a serious candidate for the application in ASSBs.
Recent Posts
-
Nanocomposites in Food Packaging
The utilization of nanocomposites in food packaging represents a significant advancement in the fiel …19th Apr 2024 -
What is the Difference Between 7075 and 6061 Aluminum Alloy?
When comparing 7075 aluminum alloy to 6061 aluminum alloy, it's essential to understand their disti …5th Apr 2024 -
Iron-Air Batteries: The Ultimate Guide
Iron-air batteries represent a significant breakthrough in energy storage technology, offering a sus …29th Mar 2024





